Application of Transcranial Direct Current Stimulation in Sleep Disturbances
Article information
Abstract
Sleep disturbances are common across all age groups, and they encompass a broad range of impairments of daytime functioning and comorbid various clinical conditions. However, current treatment methods for sleep disturbances have several limitations. As the ‘top-down’ pathway is known to play an important role in sleep-wake regulation, and as neuronal activity abnormalities have been reported as a potential pathological mechanism of sleep disturbances, the use of non-invasive brain stimulation—such as transcranial direct current stimulation (tDCS) in treating sleep disturbances—has emerged. In the present review, we first explain the mechanism of tDCS, and we also introduce recent studies that have applied tDCS to sleep disorders, along with other sleep-related tDCS studies. In conclusion, many studies have achieved improvements in sleep state, although some of these studies have reported inconsistent effects of tDCS according to the protocol and the conditions used. Further studies are needed to explore the optimal protocols to use when applying tDCS in each sleep disturbance and to enhance the evidence on the clinical efficacy of tDCS.
INTRODUCTION
Sleep is known to be a key factor for adaptation to the environment and survival [1]. Acute sleep deprivation and chronic sleep restriction impair alertness, vigilant attention, learning, memory retention, mood, and motivation [2]. Sleep disorders also negatively affect some brain pathologies, and sleep disturbances are commonly comorbid with many medical and neurological problems, including neurodegenerative disorders [3]. According to Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition (DSM-5), sleep disorders encompass various types of disturbances related to the patient’s dissatisfaction regarding the quality, timing, and amount of sleep, and they lead to daytime distress and impairment [4]. Sleep disturbances are common across all age groups [5]. Insomnia is the most common sleep disorder, and the prevalence of chronic insomnia is approximately 10%; however, the one-year incidence rate of simple insomnia symptoms is substantially higher (30%–35%) [6,7]. In the US, the total direct and indirect costs associated with insomnia have been estimated to be as high as 100 billion dollars per year [8]. Moreover, insomnia exacerbates and increases the risk of not only many neuropsychiatric disorders such as depression and dementia [9-12] but also various medical disorders, including cardiovascular [13,14], endocrine [15,16], and immunological diseases [17,18]. Sleep disturbance has also recently been shown to be a risk factor for suicide [19-21]. Therefore, the importance of its treatment has been increasingly highlighted. In general, pharmacological treatment and cognitive behavioral therapy (CBT) are used for the treatment of insomnia in general. However, medications for insomnia may have side effects, including drowsiness, dizziness, and impaired cognitive function, so the use of medication for insomnia may require special attention [22-24]. CBT requires skilled specialists and periodic hospital visits, so it is not easy to use in daily medical environments [25,26]. In addition, even with those therapies, insomnia persists in approximately 30% of patients [27]. Therefore, it might be meaningful to find an alternative treatment for sleep.
Recently, non-invasive brain stimulation methods have come to largely be used as alternative or adjunctive treatment for many neurological and neuropsychiatric disorders [27]. Among them, transcranial direct current stimulation (tDCS) is emerging in a broad range of research fields, as it is able to non-invasively and rapidly modulate brain functions such as memory, and as it has been applied as a potential treatment for various neuropsychiatric disorders over the past few decades. tDCS is the technique that changes neuronal membrane potential by flowing subthreshold direct current (1–2 mA) to the brain through an electrode attached to the scalp [28,29]. Anode stimulation induces depolarization of neurons to promote neuronal activity, and cathode stimulation induces hyperpolarization of neurons to suppress neuronal activity [30,31]. As neuronal activity abnormalities have been reported as a major pathological mechanism of sleep disorders [32], there has been an increase in studies therapeutically using tDCS for sleep disorders. Therefore, we review studies that have applied tDCS to sleep disorders, and we also examine other sleep-related tDCS studies.
MECHANISM OF TRANSCRANIAL DIRECT CURRENT STIMULATION
tDCS is one of the transcranial electrical stimulation techniques that involves the application of weak direct current that is less than the current used when delivering electroconvulsive therapy or repetitive transcranial magnetic stimulation to the scalp through two or more electrodes [33,34]. In general, the device used for tDCS consists of a current generator and two electrodes, an anode and a cathode [35,36]. The anode is the electrode through which current enters the body, and the cathode is the electrode through which current exits the body.
The current generated by tDCS is weak, and because of the resistance of living tissues such as the scalp and skull, only about 10%–20% of the current flowing to the scalp reaches the cerebral cortex and changes the activity of cranial nerves [37]. It is known than this transmitted current does not induce an action potential in the neuron, but it instead regulates the resting membrane potential of the neuron and is thus in an alteration of the neuron’s excitability, thereby changing the spontaneous discharge rate of the neuron and N-methyl-D-aspartic acid (NMDA) receptor activation [38,39].
The anode is the electrode through which current enters the body, and the cathode is the electrode through which current exits the body. Stimulation of the anode electrode hyperpolarizes the apical dendrite of pyramidal neurons of the cerebral cortex at the stimulation site, and it depolarizes soma, which changes the resting potential of the cerebral cortex close to the action potential, thus increasing neuronal activity. Conversely, stimulation of the cathode electrode hyperpolarizes the soma of pyramidal neurons in the cortex under the electrode and depolarizes the apical dendrite, thus inhibiting the resting potential from moving away from the action potential, and thereby reducing the neuronal activity in the stimulation region [34,35,39,40].
In addition, studies have shown that the effects of tDCS seem to last beyond the acute alteration of the membrane potential and may last more than several hours depending on the stimulation parameters [35,39-43]. Several studies have shown that the anode electrode stimulation of tDCS activates NMDA receptors and decreases neurotransmission by the inhibitory neurotransmitter gamma-aminobutyric acid (GABA) [35,40]. This process is considered to be related to the long-term potentiation of neuroplasticity in which the effect of tDCS is long-lasting [44]. It has also been reported that cathode electrode stimulation inhibits neuronal activity by interfering with the action of glutamate. Moreover, tDCS may affect the regulation of the acetylcholine, serotonin, and dopamine systems [35,40].
In addition to the above, the effect of tDCS has also been reported by brain model simulation [45]. Brain model simulation uses a finite element method that enables the modelling of complex brain structure and tissue characteristics that are similar to those of a real brain, and it confirms the effect of the electricity applied to the scalp on the cerebral cortex [46]. Since the white matter acts as a wire through which signals are transmitted between neurons, it becomes a path through which current flows when tDCS is applied. According to the results of studies with diffusion tensor imaging and brain model simulation, the anisotropy of white matter substantially affects the diffusion and stimulation effect of direct current stimulation, so it is presumed that tDCS activates the interaction between neurons through white matter, thereby having a stimulating effect on the brain [39,47].
APPLICATION OF TRANSCRANIAL DIRECT CURRENT STIMULATION IN SLEEP DISTURBANCES
The ‘bottom-up’ pathway has continually been shown to be important in sleep-wake cycle regulation. The ascending reticular activation system originates in the brain stem and aminergic, cholinergic, and GABAergic neurotransmission project via thalamocortical neurons into the cortex [48-51]. Pharmacologic treatment of sleep disturbances primarily targets this ‘bottom-up’ pathway; however, it is associated with limitations in terms of adverse effects and treatment efficiency [52]. Recently, an approach targeting the ‘top-down’ pathway of sleep-wake regulation with non-invasive brain stimulation has been emerging. The cortico-thalamocortical feedback loop plays an important role in regulating arousal and sleep [49,53]. Cortical neurons and synchronized slow activity underlying the occurrence of consolidated sleep serve as the primary oscillators of the cortico-thalamo-cortical feedback loop [50,54,55]. Regional synchronization of neural activity [56] and reduced metabolism in the prefrontal cortex [57] are the characteristics of sleep. The frontal cortex has been identified as being important for sleep induction and maintenance [58]. Increased electroencephalogram (EEG) beta activity during non-rapid eye movement (NREM) sleep reflecting elevated metabolism in the prefrontal cortex is a marker of arousal and the subjective perception of poor sleep [57,59].
Transcranial direct current stimulation and slow wave activity
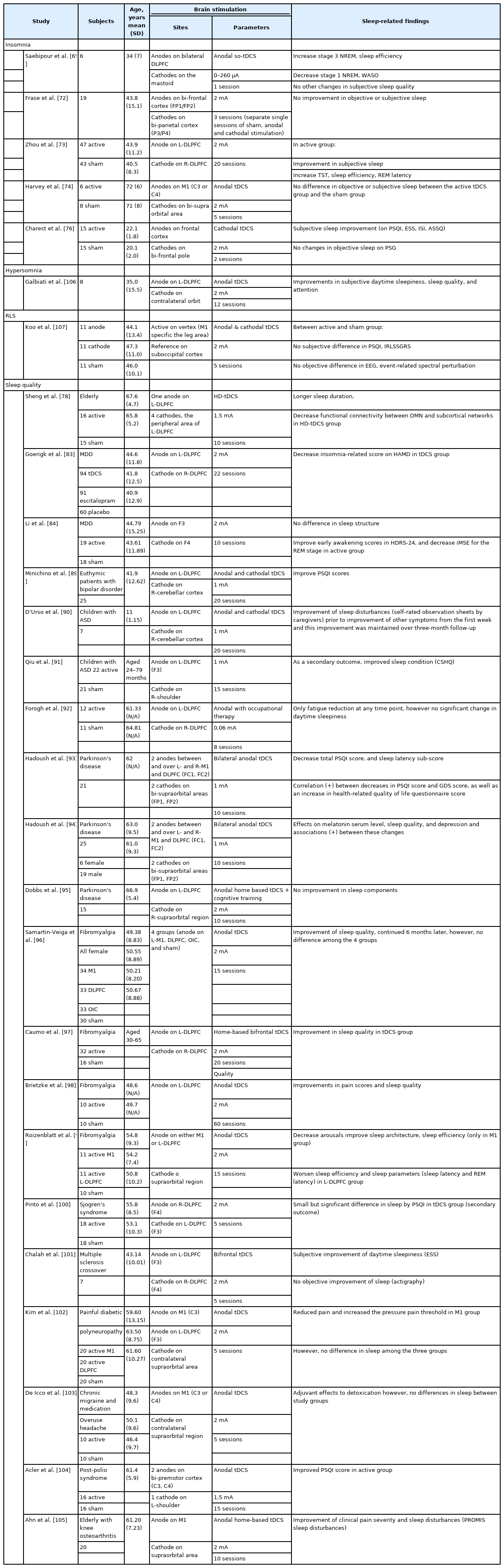
During NREM sleep, the neuronal membrane potential exhibits broadly synchronized slow subthreshold oscillations in the cerebral cortex, and the oscillations are revealed as consisting of slow-wave activity (SWA) in EEG [60]. SWA (0.5–4.0 Hz) is considered to be a marker of restorative function and related to beneficial functions of sleep, such as learning, cognitive performance enhancement, and mood stabilization [61,62]. Since SWA is initiated in the cerebral cortex [63], it can be induced in a top-down manner by stimulating the cortical regions of the sleep regulation system. Therefore, an increasing number of studies has investigated the effects of electric current inducing potential oscillation on the activity of cortical networks and sleep. It was also hypothesized that a weak current could similarly make or promote the activity of the sleep spindle and slow oscillation, as well as sleep-dependent memory consolidation [1,51,64,65]. Slow oscillatory tDCS (so-tDCS) studies have been conducted based on this hypothesis. However, some recent studies have indicated that so-tDCS does not improve memory consolidation despite increasing SWA and spindle activity [66-69]; however, any definite conclusions could be controversial and there is therefore a need for further refinement [70,71]. Saebipour et al. [65] conducted a study of the application of so-tDCS (0.75 Hz) with an intensity ranging from 0 to 260 μA during stage 2 of NREM sleep in six patients with either sleep maintenance or non-restorative sleep insomnia with the goal of resonating their brain waves to the frequency of sleep slow-wave. Anodes were placed on F3 and F4, and cathodes were placed on the mastoids. As a result, so-tDCS significantly increased stage 3 NREM and significantly decreased stage 1 NREM and wake time after sleep onset (Table 1). Although so-tDCS showed a beneficial effect on sleep, there are some difficulties involved in applying tDCS during sleep, so studies have also been conducted to examine the effect of daytime tDCS on sleep disturbances.
Application of transcranial direct current stimulation in insomnia
Patients with insomnia have been shown to exhibit increased cortical excitability; therefore, the modulation of cortical excitability is an interesting approach to managing insomnia [31,32]. Frase et al. [50] conducted separate single sessions of sham stimulation, anodal stimulation, and cathodal stimulation over the bi-frontal (FP1/FP2) target electrodes and the bi-parietal (P3/P4) return electrodes at the intensity of 2 mA on healthy adults without sleep disturbance. Moreover, PSG and resting EEG were measured after each stimulation session. As a result of this study, total sleep time (TST) and sleep efficiency were significantly decreased after the anodal stimulation, in comparison to the sham stimulation, and there was no significant change observed after cathodal stimulation. However, because of the ceiling effect in good sleeper, a potential effect of cathodal stimulation might have been missed. Still, after 3 years, the same researchers conducted a study on patients with insomnia using the same protocol, and they did not observe any changes in objective or subjective sleep [72]. It is presumed that healthy adults without insomnia could respond to excitatory stimulation, as cortical activity is not so high, while patients with insomnia already had sufficient cortical hyperactivity not to respond to anodal tDCS [31,51]. Therefore, the development of applicable tDCS protocols according to cortical activity levels will need to be considered in future studies. Zhou et al. [73] conducted a randomized, double-blinded controlled study on adults with insomnia and major depressive disorder. tDCS treatment was applied with the anode placed left dorsolateral prefrontal cortex (L-DLPFC) and the cathode placed right dorsolateral prefrontal cortex (R-DLPFC) at an intensity of 2 mA for 20 sessions. Compared to the sham tDCS group, the active tDCS group showed improved sleep in the form of decreased Pittsburgh Sleep Quality Inventory (PSQI) total scores, along with increased TST, sleep efficiency, and rapid eye movement (REM) latency. Compared to the results of these studies, it may be necessary in future studies to explore if more sessions using a similar protocol could induce more effective results.
Harvey et al. [74] conducted a randomized, parallel, double-blind, sham-controlled study on elderly individuals suffering from chronic pain and insomnia. Either the anodal tDCS over primary motor cortex (M1) (C3 or C4) at an intensity of 2 mA or sham tDCS for 5 consecutive days was applied. There was no observed difference between the active tDCS group and the sham group in terms of objective or subjective sleep. Jun et al. [75] performed a randomized, double-blind, sham-controlled study on seven patients with primary chronic insomnia. Subjects were treated with anodal stimulation (n=3), cathodal stimulation (n=2), or sham tDCS (n=2) over R-DLPFC (F8) target electrode and left acromion reference electrode at an intensity 2 mA. The respective sessions were conducted daily between 5 Pm and 7 Pm for five consecutive sessions. Although there is a limitation in the ability to evaluate the effect with a small number of patients, the anodal simulation group showed a tendency toward increased TST, decreased sleep latency, and increased sleep efficiency, and the cathodal and sham groups showed no difference. Sleep questionnaires did not find any improvement in subjective sleep. Charest et al. [76] performed a randomized, single-blinded study on healthy young adults (student-athletes) with insomnia. Bi-frontal cathodal stimulation was executed at an intensity of 2 mA for two sessions. In this study, the results only showed subjective sleep improvement on the questionnaires (PSQI), Epworth Sleepiness Scale (ESS), Insomnia Severity Index (ISI), and the Athlete Sleep Screening Questionnaire (ASSQ); however, there was no objective improvement on polysomnography (PSG) (Table 1). Moreover, Etoom et al. [77] plans to conduct a large, prospective, double-blinded randomized controlled trial to assess the effect of a bilateral anodal tDCS protocol in sleep management among athletes with sleep disturbances. That study plans to recruit participants from May 2022 to December 2023. Two anodes will be placed over R- and L-DLPFC (F4 and F3) and two cathodes (reference electrodes) will be placed over the left and right supraorbital areas (FP1 and FP2), and tDCS will be applied with an intensity of 1.5 mA six times over two weeks.
Application of transcranial direct current stimulation on sleep quality acompanying other clinical conditions
here are various forms of evidence estimating that sleep disturbances accompanying other clinical conditions may be associated with disturbances of cortical and subcortical pathways [49,51]. There have been various studies involving patients with various psychiatric, neurological, or medical diseases, or the elderly, despite not targeting insomnia patients. Sheng et al. [78] applied the high-definition tDCS (HD-tDCS), using many specialized compact scalp electrodes to supply current for more spatially selective stimulation in healthy older adults without any neurological and psychiatric disorders. This HD-tDCS protocol consisted of one anode on L-DLPFC (F3) and four cathodes on the peripheral area of L-DLPFC at an intensity of 1.5 mA intensity, and it was conducted over 10 sessions. Participants were assessed through resting-state functional magnetic resonance imaging (resting state fMRI) and several sleep questionnaires such as PSQI. As a result, sleep duration was found to be longer and functional connectivity between the default mode network (DMN) and subcortical networks was found to be decreased in the HD-tDCS group. The results of recent studies showed that relatively lower connectivity within DMN is associated with better sleep quality [79,80] and that the subcortical network also mediates promoting sleep by reducing the inhibitory modulation of the arousal system and regulating sleep depth [81,82]. Therefore, the results of this study suggest that the decoupling connectivity of the DMN-subcortical network is positively related to sleep changes in older adults. Moreover, most studies examining patients with other accompanying conditions used a similar stimulation site and similar protocols of tDCS on insomnia patients to confirm the improvement of sleep quality; however, they also used slightly different protocols for the characteristics of each disease. There have also been a number of studies that have derived sleep quality as a secondary outcome despite not using it as a primary outcome.
Psychiatric disoreders
Goerigk et al. [83] randomized patients with major depressive disorder into an escitalopram group, a tDCS group, and a placebo group. In that study, tDCS was applied by placing the anode on L-DLPFC and by placing the cathode on R-DLPFC at 2 mA for 22 sessions. As a result, the escitalopram group showed no significant difference in the score of insomnia-related questions on the Hamilton Depression Rating Scale (HAM-D), while the tDCS group showed a significant decrease in that score compared to the corresponding score of the placebo group. In another study, Li et al. [84] aimed to determine the effects of daytime tDCS on sleep EEG in patients with major depressive disorder. Ten sessions were conducted over 2 weeks with anodal stimulation over F3 and cathodal stimulation over F4 at 2 mA intensity. PSG was used to record sleep EEG signals, and the sleep EEG signals were analyzed using the intrinsic multi-scale entropy (iMSE) method. There was no difference in sleep structure; however, the active tDCS group showed some significant differences such as improved early awakening scores in the Hamilton Depression Rating Scale (HDRS-24) and significantly decreased iMSE after tDCS active stimulation for the REM stage. REM sleep disturbances are considered to be characteristic sign of depression. It is known that REM sleep latency decreases and that the proportion of REM increases in depressed patients [85,86], and that high complexity of sleep EEG is related with poor sleep quality. Entropy indicates the complexity of the EEG signals [87,88] (Table 1).
Minichino et al. [89] aimed to improve the sleep quality of euthymic patients with bipolar disorder by tDCS with the anode placed over L-DLPFC (Fp1) and the cathode placed over the right cerebellar cortex at an intensity of 2 mA for 15 daily sessions for 3 weeks. PSQI scores after tDCS treatment were significantly improved compared to baseline. As both the prefrontal cortex and the cerebellum are known to play a role in regulating sleep processes, the stimulation protocol of this study could be presumed to modulate prefrontal-thalamic-cerebellar circuits while improving sleep quality (Table 1).
D’Urso et al. [90] conducted a tDCS study that considered children with autism spectrum disorder. Similar to the studies by Minichino et al. [89], the cathode was placed on the right cerebellar hemisphere (corresponding to the cerebellar lobule VII on the scalp) and the anode was placed on L-DLPFC (F3). All participants underwent 20 daily consecutive sessions of 1.0 mA. According to the self-rated observation sheets completed by caregivers, improvement of sleep disturbances was observed prior to improvement of other symptoms from the first week of tDCS, and this improvement was maintained over 3-month follow-up. Qiu et al. [91] investigated the modulation effect of 15 sessions delivered over 3 weeks of tDCS with the anode placed at L-DLPFC (F3), and the cathode placed at the right shoulder at 1 mA intensity in children with autism spectrum disorder. As a secondary outcome, sleep condition, as measured by Children’s Sleep Habits Questionnaire, was found to be significantly improved compared to the sham group (Table 1).
Parkinson’s disease
In Parkinson’s disease, both L- and R-DLPFC, supplementary motor area, premotor area, and M1 are all involved in sleep functions [92,93]. Forogh et al. [92] performed a randomized, double-blind, sham-controlled parallel study considering patients with Parkinson’s disease. In the tDCS group, the anode was placed over L-DLPFC (F3) and the cathode was placed over R-DLPFC (F4) with a current intensity of 0.06 mA/cm2 for eight sessions in 2 weeks, and all subjects engaged in occupational therapy just after each session. Two questionnaires about fatigue and daytime sleepiness were checked just before/after treatment and with a 3-month follow-up. This study only showed a significant effect on fatigue reduction at any time point, however no significant change in daytime sleepiness. In 2018, Hadoush et al. [93] also conducted a study involving patients with Parkinson’s disease. tDCS had two anodes set over the left FC1 and right FC2, which are between and over the left and right M1 and DLPFC to stimulate these both areas, and two cathodes over the left and right supraorbital areas (FP1, FP2), respectively. Moreover, it was applied between 9 Am–12 Pm at an intensity of 1 mA for 10 sessions. In the case of this study, total PSQI score, and sleep latency domain sub-scores were all decreased significantly. It was also reported that there was a correlation between decreases in PSQI score and Geriatric Depression Scale score, as well as an increase in health-related quality of life questionnaire score. In 2021, researchers including Hadoush [94] investigated any potential changes in melatonin serum level, sleep functions, and depression in patients with Parkinson’s disease. This study used two bilateral anodes over the left FC1 and right FC2 and two cathodes over the FP1 and FP2, and it applied between 8:30–10:30 Am at the current intensity 1 mA for 10 sessions, like the former study. As the results of this study, bilateral anodal tDCS showed potential therapeutic effects on melatonin serum level, sleep quality, and depression in patients with Parkinson’s disease, as well as associations between these changes [94]. Dobbs et al. [95] performed a study with a remotely supervised tDCS protocol paired with cognitive training for patients with Parkinson’s disease. This protocol consisted of anode over the L-DLPFC and cathode over the right supraorbital region at the intensity of 2 mA for 10 sessions; however, there was no improvement in sleep components (Table 1).
Fibromyalgia
Samartin-Veiga et al. [96] aimed to establish the optimal area of tDCS by randomly assigning female patients with fibromyalgia to four groups (left M1, DLPFC, operculo-insular cortex, and sham). Patients in each group received 15 sessions of 2 mA anodal tDCS. Although an improvement of sleep quality was shown and continued 6 months later, there was no difference among the four groups. Caumo et al. [97] performed randomized, double-blind trials on female patients with fibromyalgia. The tDCS group was treated with 20-sessions of home-based tDCS with the anode placed at L-DLPFC, at an intensity of 2 mA. The tDCS group showed an improvement in sleep quality. Brietzke et al. [98] also conducted a tDCS study involving patients in fibromyalgia. Anodal stimulation was applied by the anode placed over L-DLPFC at an intensity of 2 mA for 60 sessions. There were significant improvements in pain scores and sleep quality. Roizenblatt et al. [99] investigated whether tDCS treatment is associated with changes in sleep structure in fibromyalgia. Patients were randomized into the sham group and groups with tDCS over M1 (M1 group) and tDCS over L-DLPFC (DLPFC group). For the M1 group, the anode was placed on M1 (C3) and the cathode was placed on the contralateral supraorbital area, and for the DLPFC group, the anode was placed on L-DLPFC (F3) and the cathode was placed on the contralateral supraorbital area. This study protocol consisted of 15 sessions conducted at an intensity of 2 mA. Sleep efficiency and sleep parameters such as sleep latency and REM latency worsened after L-DLPFC stimulation. Conversely, only M1 stimulation significantly reduced arousal and significantly improved sleep architecture, sleep efficiency, and pain (Table 1).
The other conditions
Pinto et al. [100] conducted a study about applying tDCS to treat fatigue in patients with Sjogren’s syndrome. The settings of tDCS consisted of five consecutive sessions with the anode placed over R-DLPFC (F4) and the cathode placed over L-DLPFC (F3) at an intensity of 2 mA. Among the secondary outcomes, sleep showed a small but significant difference in the tDCS group by PSQI (Table 1).
Chalah et al. [101] conducted a pilot randomized sham-controlled crossover study involving patients with multiple sclerosis. The subjects were treated by tDCS at an intensity of 2 mA over five daily sessions with the anode placed over L-DLPFC (F3) and the cathode placed over R-DLPFC (F4). This study resulted in a subjective improvement of daytime sleepiness according to the ESS, but there was no objective improvement of sleep measures checked by actigraphy (Table 1).
Kim et al. [102] investigated the effect of tDCS in patients with painful diabetic polyneuropathy. All participants were divided into three groups: the M1, DLPFC, and sham groups. Similar to the protocol of a former study [99], for the M1 group, the anode was placed on M1 (C3) and the cathode was placed on the contralateral supraorbital area, and for the DLPFC group, the anode was placed on L-DLPFC (F3) and the cathode was placed on the contralateral supraorbital area. tDCS protocol consisted of five sessions at an intensity of 2 mA. As a result, tDCS over the M1 reduced pain and increased the pressure pain threshold significantly, versus the DLPFC and the sham group. However, there was no significant change in sleep quality as assessed by TST and number of awakenings among the three groups (Table 1).
De Icco et al. [103] aimed to evaluate the effects of five daily sessions of anodal tDCS over M1 (C3 or C4) contralateral to the prevalent migraine pain side at 2 mA intensity, by considering patients with chronic pain and medication overuse headache. In the results, tDCS showed adjuvant effects to detoxication in the management of patient with chronic migraine and medication overuse headache; however, there were no significant differences in sleep condition between study groups (Table 1).
Acler et al. [104] conducted a study considering patients with post-polio syndrome. In this study, tDCS was set with two anodes on the right and left premotor cortex and one cathode above the left shoulder at an intensity of 1.5 mA for 15 sessions. After tDCS was applied, the PSQI score was improved significantly compared to the sham group (Table 1).
Ahn et al. [105] conducted a home-based self-administered tDCS study in older adults with knee osteoarthritis. Anodal home-based stimulation was applied by the anode over M1 and cathode over supraorbital area at a 2 mA intensity for 10 sessions. Upon completion of the tDCS sessions, clinical pain severity and sleep disturbances were improved (Table 1).
As such, in studies with chronic pain and movement disorder such as Parkinson’s disease and fibromyalgia, M1 and DLPFC were often targeted. Although there were some exceptions, such as no difference between each target group [96] or only improvement in symptoms other than sleep [103], related studies with single or co-targeting M1 and applied more sessions of tDCS than ten, reported sleep improvements in general (Table 1).
Application of transcranial direct current stimulation in hypersomnia
The pathophysiology of hypersomnia is associated with decreased cortical excitability [31]; therefore, Galbiati et al. [106] applied anodal tDCS over L-DLPFC and cathodal electrode over the contralateral orbit for a total of 12 sessions over 4 weeks to patients with idiopathic hypersomnia. They found improvements in subjective daytime sleepiness, sleep quality, and attention (decreased reaction times with Attentional Network Task) (Table 1).
Application of transcranial direct current stimulation in restless leg syndrome
In patients with restless leg syndrome (RLS), EEG spectral analysis of waking-resting conditions revealed increased high beta band power. This result might reflect a hyperexcitable sensorimotor cortex and an impairment of intracortical inhibitory pathways, which represent the pathophysiology of RLS [31,32]. Koo et al. [107] conducted a double-blind, randomized, sham-controlled, three-arm proof-of-concept clinical trial involving RLS patients. The subjects had the primary motor cortex (particularly the leg areas) stimulated at an intensity of 2 mA for a total of five daily sessions with the active electrode placed on the vertex (Cz) and the reference electrode placed over the suboccipital region. Stimulation was administered between 5 Pm–7 Pm. As a result, there was no difference in the scores of PSQI and the International Restless Legs Syndrome Study Group Rating Scale, or in the objective neurophysiological evidence of cortical excitability (electroencephalography data; event-related spectral perturbation) (Table 1).
CONCLUSIONS
To this point, to investigate the applicability of tDCS for sleep disturbances, the results of studies examining the mechanism of tDCS and the effects of tDCS on sleep disturbances have been reviewed. There is accumulating initial evidence that tDCS can be effective in improving various sleep disturbances and sleep quality as well as neurophysiological evidence for the action of tDCS. In addition, tDCS has the advantages of being portable, simple to operate, and relatively safe, in terms of mechanical characteristics, and it may be applied to patients with a wide range of neurological, medical, and psychiatric conditions in which sleep is deteriorated, to improve other comorbid symptoms as well as sleep problems. However, despite these evidences and advantages, caution should be taken when interpreting these studies results. There have only been a few studies performed according to each disease and condition; heterogeneous tDCS protocols were applied to each study, several studies were conducted with a small number of subjects, and some of the studies were conducted using uncontrolled, quasi-experimental methods. In the future, it will be necessary to find the optimal tDCS protocols and parameters for each sleep disorder, and there will be a need for large-scale, well-controlled studies to demonstrate repeated and consistent results on each sleep disorder and each protocol.
Notes
Funding Statement
None
The authors have no potential conflicts of interest to disclose
Availability of Data and Material
Data sharing not applicable to this article as no datasets were generated or analyzed during the study.
Author Contributions
Conceptualization: Young-Ji Lee. Data curation: Young-Ji Lee. Methodology: Dongyun Lee. Investigation: Young-Ji Lee. Software: Young-Ji Lee. Validation: Cheol-Soon Lee. Writing—original draft: Young-Ji Lee. Writing—review & editing: Bong-Jo Kim, Cheol-Soon Lee, Boseok Cha, So-Jin Lee, Jae-Won Choi, Eunji Lim, Nuree Kang, Dongyun Lee.