|
 |
- Search
| Chronobiol Med > Volume 5(1); 2023 > Article |
|
Abstract
Objective
Methods
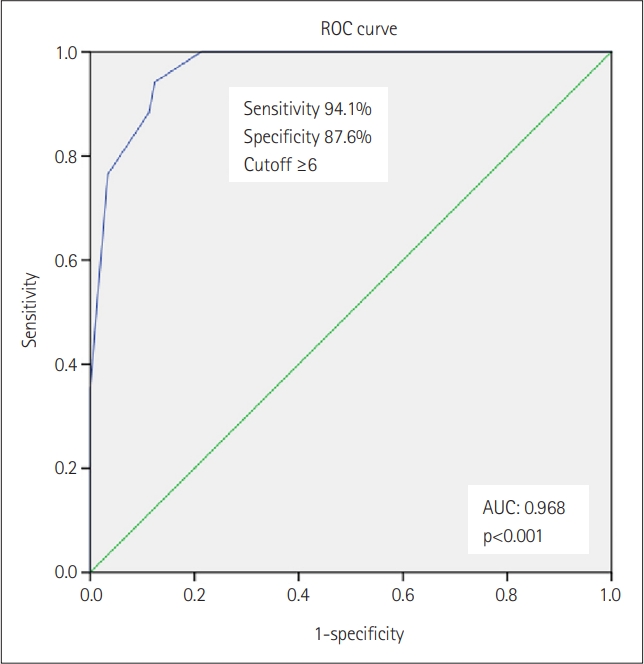
Results
Supplementary Materials
Supplementary Materials
NOTES
Conflicts of Interest
Availability of Data and Material
The datasets generated or analyzed during the study are available from the corresponding author on reasonable request.
Author Contributions
Conceptualization: Pelin Unsal, Gozde Sengul Aycicek, Burcu Balam Dogu. Formal analysis: Cafer Balci, Meltem Koca, Yelda Ozturk, Ilker Boga, Suna Burkuk, Erdem Karabulut. Investigation: Cafer Balci, Meltem Koca, Yelda Ozturk, Ilker Boga, Suna Burkuk, Erdem Karabulut. Methodology: Pelin Unsal, Olgun Deniz, Mert Esme, Ayse Dikmeer. Supervision: Burcu Balam Dogu, Meltem Halil, Mustafa Cankurtaran, Fannie Onen, SHakki Onen. Writing—original draft: Pelin Unsal, Burcu Balam Dogu, Fannie Onen, S-Hakki Onen. Writing—review & editing: Pelin Unsal, Burcu Balam Dogu, Fannie Onen, S-Hakki Onen, Meltem Halil, Mustafa Cankurtaran.
Table 1.
| Characteristics | EDS (n=53) | Control (n=53) | p |
|---|---|---|---|
| Age (yr) | 74 [65–89] | 72 [65–89] | 0.033* |
| Gender, female | 30 (56.6) | 29 (54.7) | 0.845 |
| Education | 0.591 | ||
| 0–5 years | 25 (47.2) | 24 (45.3) | |
| 6–8 years | 3 (5.7) | 7 (13.2) | |
| 9–12 years | 8 (15.1) | 8 (15.1) | |
| >12 years | 17 (32.1) | 14 (26.4) | |
| Living status | 0.105 | ||
| Couple | 26 (49.1) | 32 (60.4) | |
| Family member | 16 (30.2) | 7 (13.2) | |
| Alone | 11 (20.8) | 14 (26.4) | |
| Katz ADL score | 6 [4–6] | 6 [5–6] | 0.220 |
| Lawton–Brody IADL score | 7.3 [0–8] | 7.9 [6–8] | 0.004* |
| MNA-SF | 14 [6–14] | 14 [8–14] | 0.965 |
| MMSE | 29 [18–30] | 29 [20–30] | 0.845 |
| Yesavage Depression Scale | 2 [0–11] | 1 [0–7] | 0.050 |
| Insomnia and other sleep disorders | 27 (50.9) | 17 (32.1) | 0.049* |
| Malnutrition | 4 (7.5) | 2 (3.8) | 0.678 |
| Incontinence | 21 (39.6) | 18 (34) | 0.546 |
| Polypharmacy | 28 (52.8) | 31 (58.5) | 0.558 |
| Falls | 17 (32.1) | 16 (30.2) | 0.834 |
| Osteoporosis | 18 (34) | 13 (24.5) | 0.286 |
REFERENCES
- TOOLS