|
 |
- Search
| Chronobiol Med > Volume 5(3); 2023 > Article |
|
Abstract
Objective
Our research aimed to explore the impact of chronotype and time of day on the cognitive and psychomotor performance of medical students around 25 years old. The study provides evidence that an individual’s circadian rhythm affects their daily performance and cognitive processes.
Methods
To measure selective attention and inhibition, we utilized the Stroop test, while three subtests from the fourth edition of Wechsler Adult Intelligence Scale (WAIS-IV) were employed to measure simple attention and working memory. In addition, the Leed psychomotor tester was utilized to measure perception and motor reaction time, and the morningness-eveningness questionnaire was administered to investigate students’ chronotype. Statistical analysis were based on many tests depending on the correlation types. We mainly used ANOVA repeated measures, Bonferroni post-hoc test, ANOVA I, chi-square, and Pearson correlation tests.
Results
Our findings suggest that students exhibit decreased selective attention (F=128.842b, p<0.01, η2=0.868, 95% CI=2.744–16.998) and weakened inhibition during the late afternoon (6 p.m.) when the largest margin of error was committed (1.56±2.90) compared to 10 a.m. (0.82±1.72), leading to increased difficulty resisting interference; except that the size effect was small (η2>0.13). Simple attention reaches its peak in the morning around 10 a.m., while memory performance and auditory modality of working memory reach their peaks at 10 a.m. and the end of the afternoon (6 p.m.), respectively.
During the day, the performance of the human being knows several fluctuations according to the level of awakening in its relation to the daytime. Therefore, the fluctuations of the same variable or process present a sinusoidal rhythmicity. Many scientific studies show that behavioral vigilance in students has two main peaks and two troughs, the time period of which varies according to age [1-4]. Therefore, the school rhythm is developed in such a way as to respect the rhythm of the student in relation to his or her own homeostatic and cognitive processes [5,6]. This is said to investigate the daily rhythmicity of the latter in students, who also face the polyrhythm challenges.
A study that examines circadian rhythms as they relate to cognitive processes argues that the biological clock generates changes in one or more specific brain systems that lead to changes in cognitive processes that modulate the performance of a variety of tasks [7]. It is suggested that circadian rhythms during the performance of these tasks may be regulated primarily by three cognitive processes, including attention, working memory, and executive functions [8-10].
According to Kleitman [11], the 24-hour rhythm of metabolic activity modulates brain activity, resulting in fluctuations in cognitive function. The cerebellum and other subcortical brain structures such as the basal ganglia are involved in a variety of cognitive processes [12,13]. Attention, being one of these processes, refers to the ability to interact effectively with the environment. It involves processing and selecting incoming stimuli, responding to each stimulus, and maintaining a specific and effective response for minutes or hours [14]. According to the model by Posner and Rafal [15], attention consists of four components, including tonic vigilance, compatibility vigilance, selective attention, and sustained attention. Variations in these components are an independent manifestation of the circadian clock and not merely a passive response to changes in metabolism, including body temperature [16]. Therefore, the circadian fluctuations observed in tasks such as reaction time, memory, reading comprehension, and perceptual time can be explained by the variation of attention components [7]. From its peak during the day, the rhythm of inhibition process as one of the components of the executive functions drops at night and early in the morning [17], while that of working memory reaches a peak around 8 a.m. and decreases steadily until 1 p.m. in all chronotypes. After this period, its performance improves again in the intermediate and evening profiles [18]. Regardless of the circadian preferences of these profiles, females perform significantly better than males in visuo-motor coordination, hence the effect of gender on psychomotor performance [19].
The cognitive process rhythm varies depending on various factors, including gender, age, chronotype [20], personality traits [21], physical exercise [22], as well as experiences like the COVID-19 lockdown [23] or unique circumstances such as Ramadan fasting [24]. This raises our interest in some processes’ daily variation, notably inhibition, attention, working memory, and psychomotricity in students facing a university rhythm. Our first hypothesis suggests that the fluctuation of both chrono-cognitive and psychomotor performances of university students has two peaks: at 10 a.m. (the highest) and at 6 p.m.; and two bottoms at 2 p.m. (the deepest) and at 10 p.m. The second hypothesis assumes that chronotype has an influence on these performances according to the daytime variation.
Few studies in the literature have conducted research on the daily variation of cognitive processes in students at primary or secondary school, while no study has taken university students into consideration. Rhythms’ interactivity in the university environment might be the reason. Polyrhythms are imposed on university students, which can undoubtedly affect the daily variation of any psychomotor or cognitive characteristic that makes conducting such research difficult. Nonetheless, the current study involves the application of an experimental protocol that attempts to assess the effect of daytime on medical students’ attention, inhibition, working memory, and psychomotor fluctuations. These variations would help us determine the best time of day to study at university, taking both gender and chronotype of students into account, while also keeping in mind that other variables could have an impact on both cognitive and psychomotor performance.
This study was a prospective longitudinal study design. Participants were recruited from two medical schools and were asked to participate in the study for one day. The effect of two independent variables was investigated notably daytime and chronotype on the dependent variables: attention, memory, and inhibition as cognitive parameters and perception and motor reaction time as psychomotor parameters. Data were measured four times a day (10 a.m., 2 p.m., 6 p.m., and 10 p.m.) with a 4-hour gap between each measurement.
At the beginning of the study, participants completed a survey to collect information on their demographics: age, gender, educational attainment, chronotype, sleep duration to make sure that they are not sleep-deprivated, and a series of inquiries to verify the well-being of the students and ensure they are not afflicted by any illnesses. Cognitive and psychomotor parameters were measured using standardized tests and questionnaires. Specifically, cognitive performance was assessed using the digit span test, the letter-digit sequence, and the Stroop test, while psychomotor performance was assessed using the Leeds psychomotor tester.
In June 2021, a study was conducted to measure the daily rhythmic variation of psychomotor and cognitive processes among 62 medical students aged 25.1±0.62 years old (30 of which belong to general medicine in Casablanca), with 32 of the total sample being females. These students were in their 5th year of medical studies and were from two different institutions: the dentistry faculty of Rabat and the general medicine faculty of Casablanca. All students had internships and night shifts but had no history of sleep disorders or medication that could affect cognitive or psychomotor performance.
The dentistry students had face-to-face studies for 2 consecutive weeks followed by 2 weeks of rest, with an hourly volume of 5 to 7 hours per day, while general medicine students had an hourly volume of 8 hours, with night shifts in the hospital. Despite differences in study time, all students were invited to take the tests under the same conditions to reduce experiment bias. However, a larger sample size (351 medical students) is required to get more robust statistical findings.
In order to measure the daily variations of the cognitive processes of our sample during their academic day, we prepared an experimental protocol that consists of taking these measurements during four sessions in four periods per day; as a constant routine protocol [25]. Notably, in the morning at 10 a.m., the early afternoon at 2 p.m., the late afternoon at 6 p.m., and the late evening at 10 p.m. We called students separately in 15 small groups of four to five people (one group per day). They had to be present at 9:30 a.m., 1:30 p.m., 5:30 p.m., and 9:30 p.m. in a quiet, isolated, and well-ventilated room with constant ambient lighting to offer all the necessary conditions to make the tests. The protocol was planned in 15 different days respecting the availability of the sample from one side and the feasibility of the experiment from another. The tasks for the tests were explained before the first session in the experiment, which made it easier to make them for the three other sessions. The students were better situated and knew exactly what they had to do on each task.
In order to obtain accurate measurements, we utilized the most dependable tools that were selected based on their psychometric dimensions. These tools enabled us to conduct unbiased analyses, accounting for any potential bias that may have been present during the experimental protocol. Prior research has employed the same tools to assess both psychomotor and chronocognitive performance, which refers to daily evaluations of cognitive processes in order to determine their patterns throughout the day. We measured these parameters four times daily and maintained a consistent testing order for all participants, culminating in a final test that gauged central nervous system (CNS) activation and evaluated their ability to resist fatigue and remain alert following completion of all tasks.
The Morningness-Eveningness Questionnaire (MEQ) is a commonly used survey that helps determine whether a person prefers morning or evening activities. Developed in 1976 by Horne and Östberg [26], the MEQ has been revised by other researchers. The questionnaire has strong internal consistency, with a Cronbach’s alpha coefficient of 0.70 to 0.86, indicating that it measures the same construct. Additionally, test-retest reliability over one month is approximately 0.75, indicating that the questionnaire is consistent over time [27,28].
We started first by the digit span subtest from the fourth edition of Weshler Adult Intelligence Scale (WAIS IV) by Wechsler, in 2008 [29]. It is one of the most used tests to measure the auditory modality of the working memory [30]. The digit span subscale includes the oral presentation of numbers to the participant at 1-second intervals, using the same voice tone all the test long. Participants must memorize as many numbers as possible in a specific order. Digit span consists of two tests: forward (DSF) and backward (DSB). In the first one, they must memorize numbers in the presented order. By contrast, in the second one, participants are asked to remember the numbers in reverse order [31]. The reliability of the internal subscale was slightly higher at 0.93, while, the mean internal consistency of the DSF and DSB test results (over the age group) were given as 0.81 and 0.82, respectively [29].
The letter-number sequencing subscale is a supplementary subtest to measure the processing speed and visual-spatial working memory components (using the visualizing numbers and letters strategy as they are placed in ascending/alphabetical order) [32]. In the WAIS IV letter-number sequencing subtest, participants are given 10 sequences of 3 training tasks and the number of tasks ranging from two to eight characters between letters and numbers [30]. It involves generally many cognitive processes such as: attention, concentration, mental manipulation, sequential processing, memory span, and short-term auditory memory [33].
The third test was the Stroop Color and Word Test (SCWT) by Stroop (1935). We have taken the Golden’s version (1978) being the most widely used [34]. Clinically, it evaluates a specific aspect of executive function, i.e., selective inhibition. This requires the subject to suppress the automatic read response instead of the less prepared competing color naming response [35]. Our sample are invited to read the words in three different panels. In the first one, subjects are asked to read words printed in black. In the second one, the same words are printed in color and subjects are asked to name the color of the ink corresponding to the name of the color itself. While in the third one, subjects are asked to name the color of the ink, which does not correspond to the word they are reading [36].
To assess the participants’ psychomotor abilities, we used the Leeds psychomotor tester notably the choice reaction time (CRT) test and the critical flicker fusion (CFF) test. The CRT test measures the speed of reaction by placing the finger on a button in order to react to a visual signal (green light) when one of the six light-emitting diodes (LEDs) lights up to measure both the recognition reaction time (RRT) and the CRT. When the CFF threshold being an indicator of alertness and mental fatigue measures visual discrimination and general arousal (the activation of the CNS). The subject was asked to distinguish the flicker; a series of flashes separated by dark intervals; in an array of four LEDs by centralizing his binocular vision and fixing it at about 1 m from the array until the distinctness of the flashes shifts to a continuous flicker [37]. The average threshold of the CFF test in our study was derived using the limit method from three presentations with increasing frequencies (CFF up) [38-40].
The study employs a repeated measures design with a multivariate approach to measure cognitive and psychomotor parameters at 4-hour intervals throughout the day using analysis of variance (ANOVA) repeated measures test (p-values, frequencies, and partial eta squared). Means and standard deviations were also calculated demonstrating the values’ degree of dispersion within our dataset. Multivariate tests notably Pillai’s trace, Wilks’ lambda, Hotelling’s trace, and Roy’s largest root test were used to consider the correlation between repeated measures and effects of independent variables on multiple dependent variables. Additionally, paired comparisons were used to compare the values of dependent variables at different times of the day within participants, and p-values were adjusted using Bonferroni correction to account for multiple comparisons, using the Bonferroni post-hoc test. For the chronotype, chi-square, ANOVA I, and Pearson correlation tests were used to measure its correlations with cognitive and psychomotor parameters during the day. The study ensures that assumptions of the tests are met, including normality and homogeneity of variance-covariance matrices.
The study followed ethical guidelines and obtained informed consent from all participants. Participants were informed about the purpose and procedures of the study and were given the option to withdraw from the study at any time without any penalty. The data collected were kept confidential and anonymous, and participants were assured that their personal information would not be shared with any third party.
The implementation of the experimental protocol to measure cognitive and psychomotor processes in relation to chronotype and daytime posed several challenges. Maintaining participant compliance was difficult, particularly with adhering to strict fourhour intervals for measurement, and some participants struggled to complete the tasks due to fatigue or other factors. Attrition was also a problem as some participants found the study tasks tedious or time-consuming. Conducting four measurements per day required significant resources and time commitment from both the researchers and participants, and not all participants were fully engaged. Out of 83 students, only 62 were able to be maintained. Lastly, there is a concern for generalizability as the study was conducted among medical students who may not represent the broader population, and the findings may not be applicable to other contexts or populations.
The results showed that gathered data satisfy assumptions of normal distribution (all p>0.05) and homogeneity of variance (all p>0.05).
At 10 a.m., the effect of the time of day on the performance of students in reading colors in raw letter (RCRL) mode and in reading colors in final letter (RCFL) mode (139.05±26.73; 138.53±26.73) was higher than at 2 p.m., a period during which their performance had decreased (117.31±26.12). According to the Bonferroni paired comparison test, the difference in the mean scores of RCRL and RCFL modes between the 10 a.m. and 2 p.m. periods is highly significant (p<0.001). Similarly, for other periods of the day, performance at 10 a.m. remains better than at 6 p.m. (F=128.842b, p<0.01, η2=0.868, 95% confidence interval [CI]=2.744– 16.998) and 10 p.m. (F=128.842b, p<0.001, η2=0.868, 95% CI=7.467–23.049) according to the ANOVA repeated measures.
However, the effect of the time of day on the average performance of RCRL and RCFL modes is more significant at 6 p.m. than at 2 p.m. (F=128.842b, p<0.001, η2=0.868, 95% CI=4.318– 19.424) and 10 p.m. (F=128.842b, p<0.01, η2=0.868, 95% CI=1.018–9.756). While the number of errors made was less prominent at 10 a.m. than at 2 p.m. when it reached its peak (0.52±1.04 vs. 1.73±3.05). Nonetheless, the effect of the time of day on the averages of student performance in RCRL and RCFL modes was highly significant (p<0.001) and the associated effect size was large (η2>0.8), based on repeated measures ANOVA analysis.
For the task of reading colors in raw color (RCRC) mode and reading colors in final color (RCFC) mode, students’ performances were approximately similar between the periods of 10 a.m., 6 p.m., and 10 p.m. (raw: 100.94±24.95; 100.65±17.24; 100±13.70 vs. final: 100.11±25.17; 99.08±17.98; 98.58±14.01, respectively). However, at 2 p.m., the raw (94.66±18.52) and final (93.39±18.88) performance of participants in this task experienced a notable decrease. However, the largest margin of error was committed at 6 p.m. (1.56±2.90) while the smallest was committed at 10 a.m. (0.82±1.72). Based on the ANOVA repeated measures, the effect of the time of day on the difference of performance at the RCRC and RCFC modes is significant (p<0.05) and the effect size is small (η2>0.13).
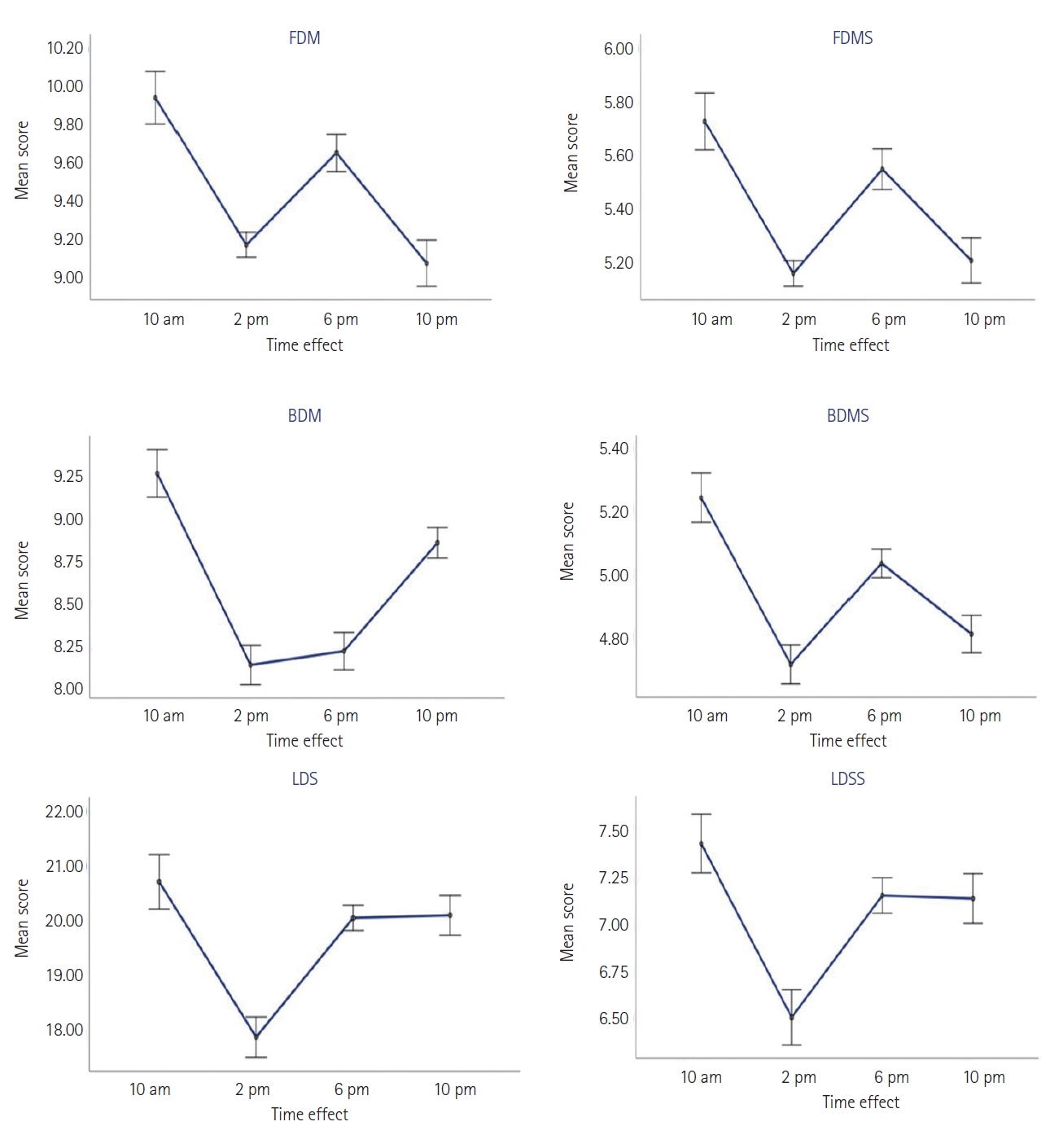
The scores for forward digit memory (FDM) and its digit span (FDMS) fluctuate throughout the day with two peaks at 10 a.m. and 6 p.m. and two lows at 2 p.m. and 10 p.m. The deepest dip in FDM occurs at 10 p.m. (9.06±0.96), while the lowest point for FDMS is in the afternoon around 2 p.m. (5.16±0.37). However, the highest peak scores for both FDM (9.94±1.08) and FDMS (5.73±0.83) coincides with the morning period at 10 a.m. The performance difference of FDM between 10 a.m. and 2 p.m., 10 p.m., 6 p.m. is highly significant (F=13.656b, p<0.001, 95% CI=0.423–1.126; F=13.656b, p<0.001, 95% CI=0.348–1.394; and F=13.656b, p<0.001, 95% CI=0.160–0.808, respectively). The difference between 6 p.m. and 10 p.m. is also significant (F=13.656b, p<0.005, 95% CI=0.148–1.013). The effect of time of day on the mean performance of FDMS is highly significant between 10 a.m. and 2 p.m.; 6 p.m. and 2 p.m.; and 10 a.m. and 10 p.m. (F=12.560b, p<0.001, 95% CI=0.280–0.84; F=12.560b, p<0.001, 95% CI=0.143–0.61; and F=12.560b, p<0.005, 95% CI=0.126– 0.90, respectively). The difference between 6 p.m. and 10 p.m. is also significant (F=12.560b, p<0.05, 95% CI=0.001–0.67). However, the effect size of these differences on FDM and FDMS performance is moderate (FDM: p<0.001, η2=0.410 vs. FDMS: p<0.001, η2=0.390).
Similarly, the backward digit memory (BDM) and letter-digit sequence (LDS) scores fluctuate with two peaks at 10 a.m. and 10 p.m. and a single dip at 2 p.m. However, their digit spans (BDMS and LDSS) show a sinusoidal variation with two peaks at 10 a.m. and 6 p.m. and two dips at 2 p.m. and 10 p.m. The highest peak for BDM (9.26±1.09), BDMS (5.24±1.09), LDS (20.71±3.92), and LDSS (7.42±6.50) is reached in the morning at 10 a.m., while the deepest dip for these variables is in the afternoon at 2 p.m. (8.15± 0.90; 4.71±0.49; 17.87±02.91; 6.50±1.16, respectively) (Figure 3). According to the repeated measures ANOVA analysis, the effect of daily time variation on the performance of BDM, BDMS, LDS, and LDSS across different periods of the day is highly significant. The effect of the size of the difference in performance levels is moderate (BDM: F=24.271b, p<0.001, η2=0.552; BDMS: F=15.826b, p<0.001, η2=0.446; LDS: F=18.303b, p<0.001, η2=0.482; LDSS: F=10.87b, p<0.001, η2=0.356) (Figure 3).
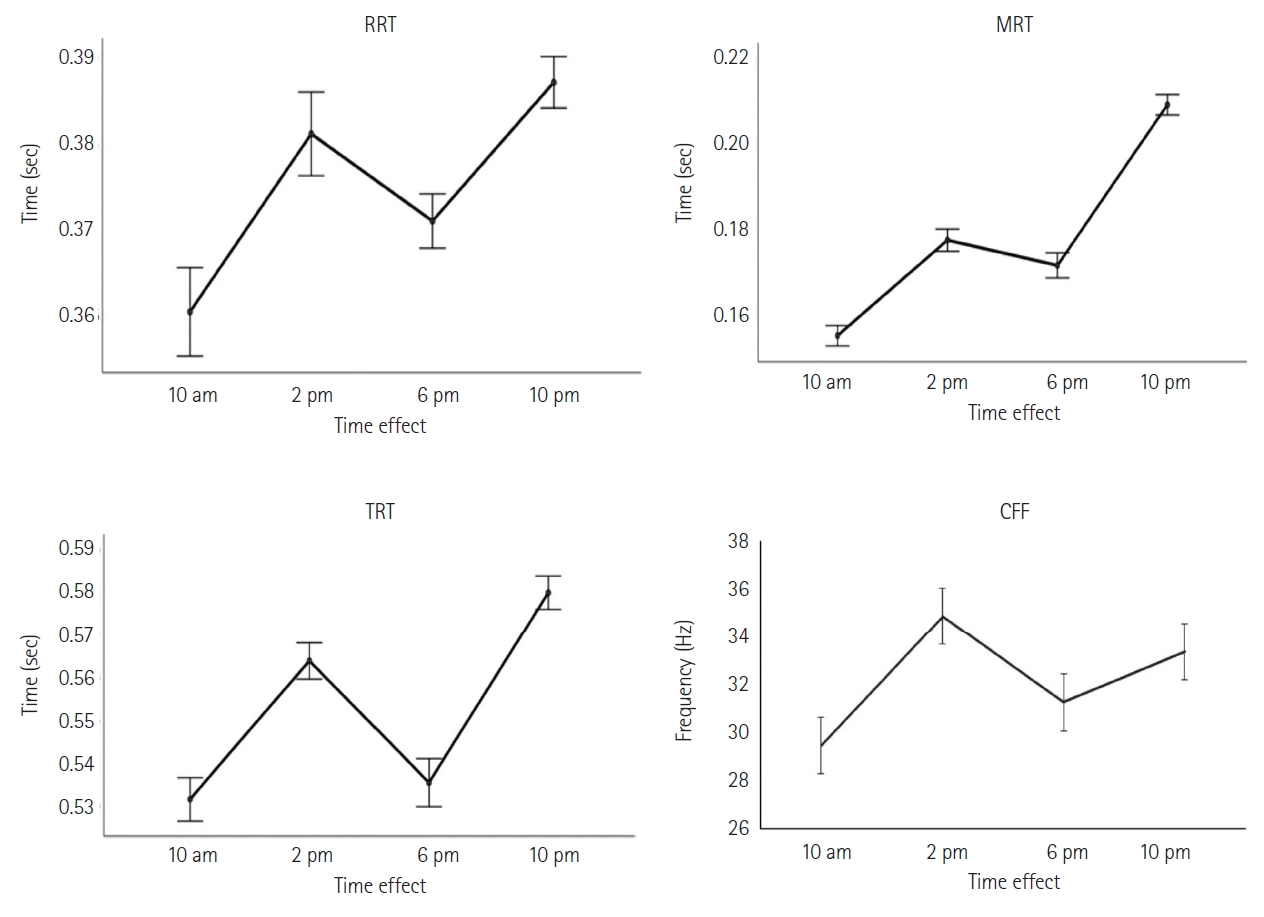
The scores on the Leeds psychomotor tester showed a sinusoidal fluctuation, but with an inverse pattern compared to the Stroop test or memory variables. There were two dips at 10 a.m. and 6 p.m., and two peaks at 2 p.m. and 10 p.m. However, the two dips represented the points where reaction speed peaked for RRT (0.36±0.04; 37±0.02), motor reaction time (MRT; 0.15±0.02; 0.17± 0.02), or total reaction time (TRT; 0.53±0.04; 0.54±0.03), while the two peaks represented the points where RRT (0.38±0.03; 0.38±0.02), MRT (0.18±0.02; 0.20±0.02), and TRT (0.56±0.04; 0.58±0.03) became much slower. Additionally, the CFF was closest to 25 Hz at 10 a.m. and 6 p.m., while it deviated further from 25 Hz at 2 p.m. and 10 p.m. (Figure 4).
According to Bonferroni’s paired comparison, the difference in mean PRT performance between 10 a.m. and 2 p.m. was highly significant (p<0.01), and between 10 a.m. and 10 p.m. was very highly significant (p<0.001), demonstrating that 10 a.m. was the time when the students achieved the fastest perception speed during the day. The MRT remained consistently better in the morning, particularly at 10 a.m., as evidenced by the highly significant differences in mean MRTs between 10 a.m. and other time periods (p<0.001). As a result, the TRT was similarly faster at 10 a.m. due to the highly significant differences in mean TRTs between 10 a.m. and 2 p.m. (p<0.001) and between 10 a.m. and 10 p.m. (p<0.001).
However, in the repeated measures ANOVA of psychomotor tests, it was shown that the effect of time of day on RRT, MRT, and TRT was highly significant (p<0.001) but with a small effect size compared to RRT (F=6.516b, p<0.001, η2=0.249), medium compared to TRT (F=27.314b, p<0.001, η2=0.581), and large compared to MRT (F=82.532b, p<0.001, η2=0.808). Meanwhile, for the general arousal test, the students were much more alert at 10 a.m. and 10 p.m., as demonstrated by the highly significant performance difference in CFF, with a medium effect size (F=32.092b, p<0.001, η2=0.620) compared to other time periods.
Compared to our sample, 29% of students are intermediate chronotypes, of which 14.5% are female; 25.8% are evening chronotypes, of which 14.5% are male; and 45.2% are morning chronotypes, of which 51.6% are female. This means that the majority of our students are morning chronotypes, especially females. However, according to the chi-square and ANOVA I tests, gender does not have a significant effect on chronotype, either in category or score (p>0.05).
The chronotype score has a highly significant, positive correlation of moderate intensity with RCRL and RCFL modes at 10 a.m. and 2 p.m. (RCRL: r=0.774, p<0.001; r=0.717, p<0.001/RCFL: r=0.774, p<0.001; r=0.716, p<0.001, respectively), and a weak intensity at 6 p.m. and 10 p.m. (RCRL: r=0.537, p<0.001; r=0.523, p< 0.001/RCFL: r=0.535, p<0.001; r=0.536, p<0.001, respectively), according to the Pearson correlation test. Similarly, a positive and highly significant correlation was established between RCRC and RCFC modes with the chronotype score at moderate intensity at 10 a.m. (RCRC: r=0.696, p<0.001/RCFC: r=0.698, p<0.001), and at a weak intensity at other times of the day. Another significant, positive, and weak intensity correlation was established between the chronotype score and inhibition at 10 a.m. (r=0.251; p< 0.05), highly significant and weak intensity at 2 p.m. (r=0.539; p< 0.001), and very significant and weak intensity at 6 p.m. (r=0.296; p<0.01) and 10 p.m. (r=0.324; p<0.01).
Regarding the correlations of memory test variables with the chronotype score, only two were significant, positive, and of weak intensity, namely BDM at 10 a.m. (r=0.264; p<0.05) and LDS at 2 p.m. (r=0.262; p<0.05). There is a highly significant and positive correlation between the chronotype score and TRT at 2 p.m. (r=0.379, p=0.01), and a significant and negative correlation between the chronotype score and TRT at 6 p.m. (r=-0.264, p<0.05). There is also a highly significant and negative correlation between the chronotype score and MRT at 10 a.m. (r=-0.609, p<0.001) and 10 p.m. (r=-0.419, p<0.001), and a highly significant and positive correlation between the chronotype score and MRT at 2 p.m. (r=0.504, p<0.001). However, there is no significant correlation between the chronotype score and RRT throughout the day.
Regarding the general state of arousal, the students’ chronotype score had a highly significant, negative correlation of moderate intensity with the frequency of CFF at 10 a.m. (r=-0.503, p<0.001) and 10 p.m. (r=-0.523, p<0.001).
We remind that our study aims to investigate two initial hypotheses: the first is that the time of day has an effect on cognitive and psychomotor performances, and the second argues that chronotype is also correlated with these same performances among medical students. Before moving on to the analysis of the results obtained, we note that daily variation is an integral part of the circadian rhythm (24 hours). According to Valdez et al. [7], the circadian rhythm of cognitive processes for individuals with an intermediate chronotype who usually sleep from 11 p.m. to 7 a.m. is low in the morning between 7 a.m. and 10 a.m., improves around noon, reaches its first peak between 10 a.m. and 2 p.m., and decreases after lunch towards its first dip between 2 p.m. and 4 p.m. Its second peak is reached in the late afternoon and early evening between 4 p.m. and 10 p.m., and gradually decreases between 10 p.m. and 4 a.m. to reach its lowest levels (second dip) at dawn and early morning between 4 a.m. and 7 a.m.
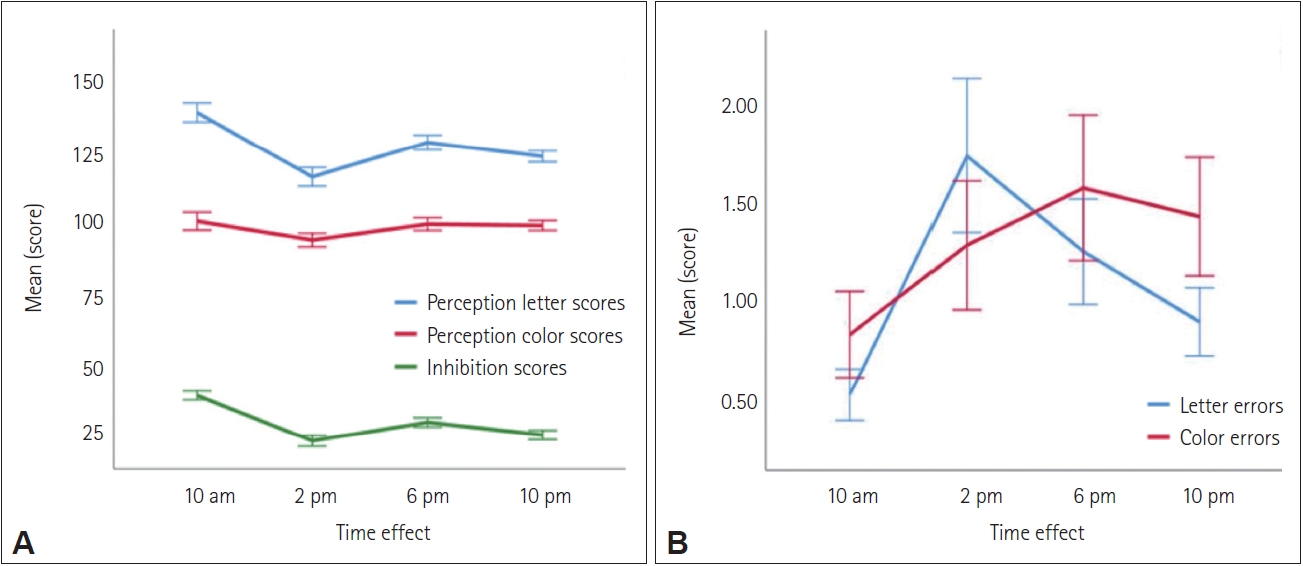
The daily performance of individuals varies depending on the time of task execution. The different processes interacting in the human brain are related on the one hand to the type and conditions of the task, and on the other hand to the brain’s temporal ability to execute it [41]. Attention, as a cognitive process, was measured in this study by the Stroop test. Our sample demonstrated variations in the tasks to be performed in the Stroop test depending on the different periods of the day, so that the mean scores of the first task (RCFL mode) were significantly higher than those of the second task (RCFC mode). However, the mean errors for the second task exceed those of the first during all periods of the day. This shows a notable decrease in perception and inhibition in students in RCFC mode compared to RCFL mode, resulting in a reduction in selective attention and an increase in the difficulty of resisting interference. These results confirm the findings of two previous studies, one of which reported that weaker inhibition was observed in the early evening [42], and the other revealed that a decrease in inhibition was observed at 1 a.m. and 7 a.m., compared to 1 p.m. and 7 p.m. [43].
Moreover, following the observation of the daily variation in error scores, we note that those committed in the RCRL mode increase at 2 p.m. compared to 10 a.m., while those committed in the RCRC mode increase at 6 p.m. compared to 2 p.m., highlighting the increased perceptual difficulty, lack of attention, and weak resistance to interference among our students in the late afternoon (6 p.m.). These findings partially converge with the conclusions of Valdez [14] in his literature review (2019), which indicate that during the early morning hours, particularly between 4 a.m. and 7 a.m., and during late hours, productivity and job performance decline, with an increased risk of errors and accidents. Additionally, the decreased attention during these times of the day also has consequences on academic learning.
On the athletic front, research has shown that self-paced athletes, such as swimmers and runners, perform better on inhibition tasks [44]. Therefore, it is preferable to train at the time of day when inhibition reaches its peak, particularly at 10 a.m. according to our study.
On the other hand, our results show that in terms of scores on the FDM and FDMS, it can be suggested that simple attention among our students reaches its highest peak in the morning around 10 a.m., then decreases towards the lowest performance in the early afternoon (2 p.m.), and reaches its second peak towards the end of the afternoon (6 p.m.), before remarkably declining at night around 10 p.m. Our study also highlighted that the time of day has an impact on memory performance, due to significant differences in the performances measured by the WAIS subtests during different periods of the day (with 4-hour gaps). This reinforces the findings of Evansová et al. [45], who found differences in the declarative memory results of participants evaluated using the Rey Auditory Verbal Learning Test task (related to the time of evaluation with a half-hour gap).
Meanwhile, in the auditory modality of working memory represented by variations in scores of BDM, LDS, and LDSS among our students, the curve plotted throughout the day showed two peaks, with the highest at 10 a.m. and a single trough at 2 p.m. This finding supports the results of Folkard [46], who demonstrated a time-of-day effect on a working memory task under daynight conditions. However, studies conducted by Johnson et al. [47] and Monk and Carrier [48] suggest that performance differences in tasks observed during normal day-night conditions may be more influenced by the homeostatic drive of wake time than by the circadian timing system (CTS). The use of a forced desynchronization protocol in these studies allowed for the distinction of the two factors. Short-term memory, verbal reasoning, and serial search were found to systematically vary with wake time and follow the rhythm of body temperature, with the influence of wake time being at least as strong as that of the CTS on these specific tasks. These results underscore the importance of considering wake time and body temperature when studying performance in different tasks [47,49].
The daily variation of TRT in our study shows two troughs, the first of which represents the fastest reaction time at 10 a.m., and two peaks, the second of which represents the slowest reaction time at 10 p.m. However, MRTs are much faster than perception reaction times in comparison to all times of the day. This means that the motor movement takes less time than the information processing that precedes perception. The MRT also suggests that the two periods around 10 a.m. and 6 p.m. are the best times for physical activity, since the MRT reaches its peak performance during the day at these times in our students. This is further reinforced by the results of the CFF in our study, which confirm that the general arousal of students is much higher during these two time slots, particularly at 10 a.m. and 6 p.m., knowing that higher CFF scores indicate greater perceptual accuracy.
According to the correlation between chronotype and the RCC mode task among our students, we can understand that the more the chronotype score increases towards morning types, the more attention increases during the execution of the Stroop test tasks compared to all periods of the day. This explains the relationship between morningness and attention. In the same vein, Evansová et al. [45] demonstrated that chronotype has a significant effect on the Stroop subtest on color naming (i.e., RCC in our study) much more in extreme morning types than in extreme evening types, while no difference was observed between extreme morning types and intermediate types.
However, the correlation between chronotype and inhibition score in the afternoon demonstrates that the more the chronotype score increases towards morningness, the more inhibition increases in our students, particularly at 2 p.m., which explains why morning types are less efficient and attentive in the afternoon. Our study also validates the effect of chronotype on memory when it correlates with both the BDM at 10 a.m. and the LDS at 2 p.m.; this suggests that students who tend towards morningness perform better in terms of digit memory in the morning and LDS memory in the afternoon. Our study converges with the results of Schmidt et al. [50] who confirmed that chronotype and time of day interact with the working memory load. A more recent study has also revealed that morning chronotypes showed positive results in all measures of cognitive and physical performance compared to late chronotypes during the morning hours, particularly at 8 a.m. [51].
Following the correlations between chronotype and vigilance factors, namely RRT, MRT, and TRT, in our study, it turns out that the visual perception speed measured via RRT in our students has no relationship with their chronotype tendencies. On the other hand, the MRT showed to be faster in students with morning tendencies, particularly in the morning at 10 a.m. and in the late afternoon at 6 p.m., unlike 2 p.m. when their MRT is much slower. Furthermore, the correlation between chronotype and TRT in our study suggests that when the students’ chronotype score tends towards morningness, their TRT becomes slow in the early afternoon (2 p.m.) and fast in the late afternoon (6 p.m.). Therefore, morning types have better motor skills in the morning at 10 a.m. and in the afternoon at 6 p.m., and better vigilance at 6 p.m. Similarly, Facer-Childs et al. [51] also found a significant correlation between time of day and chronotype for psychomotor vigilance task performance. Early chronotypes performed better in the morning than in the afternoon and evening, whereas late chronotypes showed significant variation with poorer morning performance compared to evening. The performance variation was 3.5% for early chronotypes and 9.1% for late chronotypes. At 8 a.m., early chronotypes performed significantly better (8.4%) than late chronotypes. Furthermore, the correlation between the CFF and chronotype suggests that the wakefulness state of the students is more active at 10 a.m. and 10 p.m. (the frequency of the CFF is much closer to 25 Hz, the average at which humans must perceive flickering in a fixed light), when the chronotype score tends towards morningness.
However, it should be noted that throughout our experimentation, we ran across a number of challenges that endangered the favorable settings for our research. The fact that the tests must be completed in around 45 minutes every 4 hours, for four times a day, causes students to perceive the experiment as time-consuming and difficult. In order to comply with the experiment’s requirements, students had to almost stop performing their usual everyday activities. Additionally, as we noted in the paragraph “Research difficulties,” we have encountered many other issues such as students’ availability, and their level of readiness for the experiment. For this reason, further studies with bigger populations are required to get statistical results that are more conclusive for university students in general and medical students in particular.
The study confirms two hypotheses. The first is that the time of day affects cognitive and psychomotor performance, as shown by variations in performance on the Stroop test, forward and backward digit memory, and letter digit sequence at different times of the day. The second hypothesis is that a person’s chronotype is linked to cognitive and psychomotor performance. For example, students show the highest simple attention performance in the morning at around 10 a.m. before declining in the early afternoon (2 p.m.), as it was previously shown that self-paced athletes perform better when inhibition is at its peak. Overall, the study provides evidence that both time of day and chronotype are important factors that can impact cognitive and psychomotor performance.
We can conclude that medical students’ daily studies in the university are affected by their cognitive and psychomotor daily fluctuations. The study suggests that students’ cognitive and psychomotor abilities can vary throughout the day and may be influenced by factors such as sleep, diet, exercise, and stress. Given these fluctuations, it may be beneficial for medical students to schedule their study time and activities based on their individual rhythms and preferences. For example, some students may find it more effective to study in the morning when their cognitive abilities are at their peak, while others may prefer to study in the afternoon or evening. Additionally, students may want to incorporate activities such as exercise or meditation into their daily routines to help improve their cognitive function and reduce stress.
Overall, the article highlights the importance of understanding and accounting for individual cognitive and psychomotor fluctuations when planning and implementing daily study routines for Moroccan medical students. However, further research with larger sample sizes and more precise and adapted experimental protocols is warranted.
NOTES
Conflicts of Interest
Availability of Data and Material
The data presented in this study are available on request from the corresponding author. The data are not publicly available due to confidential reasons.
Author Contributions
Conceptualization: all authors. Data curation: Ikram Sabaoui. Formal analysis: Said Lotfi. Investigation: Ikram Saboui, Said Lotfi. Methodology: Ikram Sabaoui, Said Lotfi. Project administration: all authors. Resources: Ikram Sabaoui, Said Lotfi. Software: Ikram Sabaoui, Said Lotfi. Supervision: Said Lotfi, Mohammed Talbi. Validation: all authors. Visualization: Said Lotfi, Mohammed Talbi. Writing—original draft: Ikram Sabaoui. Writing—review & editing: all authors.
Acknowledgments
The authors would like to thank all the students who contributed in the study data collection during their academic days.
Figure 1.
Daily mean score variation of Stroop tasks test. RCRL mode, reading colors in raw letter mode; RCL mode errors, reading colors in letter mode erros; RCFL mode, reading clors in final letter mode; RCRC mode, reading colors in raw color mode; RCC mode errors, reading colors in color mode errors; RCFC mode, reading colors in final color mode. Error bars: ±1 standard deviation.
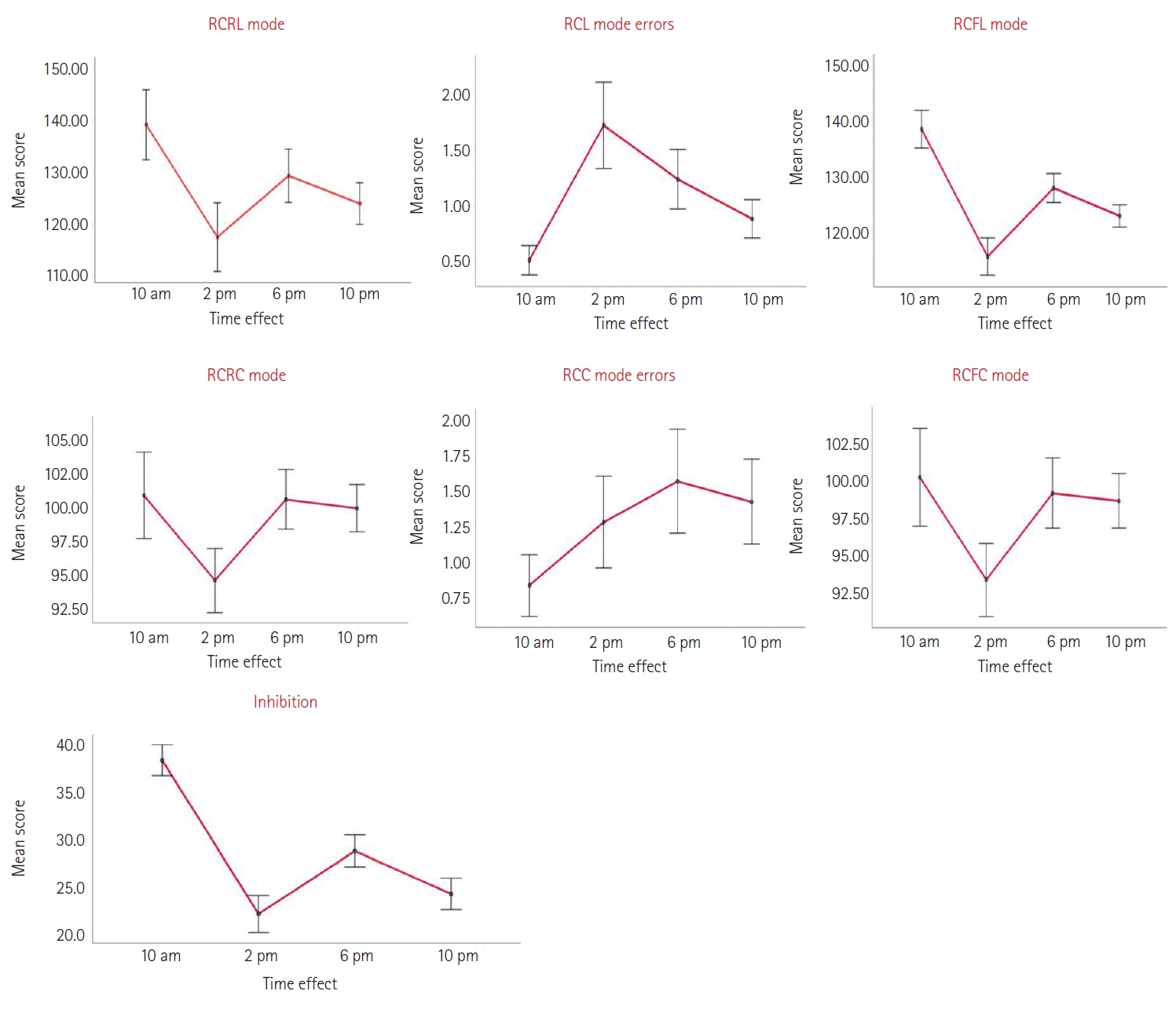
Figure 2.
Comparison of perception and inhibition (A) and reading errors (B) daily fluctuations from the Stroop test.
REFERENCES
1. Hellbrügge T. The development of circadian rhythms in infants. Cold Spring Harb Symp Quant Biol 1960;25:311–323.


2. Koch P, Montagner H, Soussignan R. Variation of behavioral and physiological variables in children attending kindergarten and primary school. Chronobiol Int 1987;4:525–535.


3. Janvier B, Testu F. [Développement des fluctuations journalières de l’attention chez des élèves de 4 à 11 ans]. Enfance 2005;57:155–170.French.

5. Jacob BA, Rockoff JE. Organizing schools to improve student achievement: start times, grade configurations, and teacher assignments. Washington, DC: Brookings, 2011.
6. Adolescent Sleep Working Group; Committee on Adolescence; Council on School Health. School start times for adolescents. Pediatrics 2014;134:642–649.



7. Valdez P, Ramírez C, García A. Circadian rhythms in cognitive processes: implications for school learning. Mind Brain Educ 2014;8:161–168.

8. Cajochen C, Khalsa SB, Wyatt JK, Czeisler CA, Dijk DJ. EEG and ocular correlates of circadian melatonin phase and human performance decrements during sleep loss. Am J Physiol 1999;277(3 Pt 2): R640–R649.


9. Schmidt C, Collette F, Cajochen C, Peigneux P. A time to think: circadian rhythms in human cognition. Cogn Neuropsychol 2007;24:755–789.


11. Kleitman N. Sleep and wakefulness. Chicago: The University of Chicago Press,, 1963, p. 140.
12. Giovannucci A, Badura A, Deverett B, Najafi F, Pereira TD, Gao Z, et al. Cerebellar granule cells acquire a widespread predictive feedback signal during motor learning. Nat Neurosci 2017;20:727–734.




13. Wagner MJ, Kim TH, Savall J, Schnitzer MJ, Luo L. Cerebellar granule cells encode the expectation of reward. Nature 2017;544:96–100.




15. Posner M, Rafal R. Cognitive theories of attention and the rehabilitation of attentional deficits. In: Meier M, Benton A, Diller L. editors. Neuropsychological rehabilitation. New York, Guilford Press, 1987, p. 182–201.
16. Wright KP, Lowry CA, Lebourgeois MK. Circadian and wakefulness-sleep modulation of cognition in humans. Front Mol Neurosci 2012;5:50.



17. García A, Ramírez C, Martínez B, Valdez P. Circadian rhythms in two components of executive functions: cognitive inhibition and flexibility. Biol Rhythm Res 2012;43:49–63.

18. Nasiri Zarch Z, Sharifi M, Heidari M, Pakdaman S. Investigating chronotype orientation on daily and weekly rhythm fluctuations in preschoolers working memory performance. Int Clin Neurosci J 2019;5:150–157.


19. Campos TF, Silva SB, Fernandes MG, Araujo JF, Menezes AA. Diurnal variation in a visual-motor coordination test in healthy humans. Biol Rhythm Res 2001;32:255–262.

20. Sabaoui I, Lotfi S, Talbi M. Analytical study of the impact of age chronotype and time preferences on the academic performance of secondary school students from a modest social background. Retos 2022;46:631–640.


21. Bouiri O, Lotfi S, Talbi M. Correlative study between personality traits, student mental skills and educational outcomes. Educ Sci 2021;11:153.

22. Lotfi S, Elmoutaraji I, Talbi M. Effect of physical exercise and gender on information processing and choice reaction time of university students. Int J Hum Mov Sports Sci 2020;8:37–42.

23. Sabaoui I, Lotfi S, Talbi M. Variations in circadian rhythmicity and students’ gender-related psychological conditions during the COVID-19 lockdown. Educ Sci 2021;11:355.

24. Lotfi S, Madani M, Tazi A, Boumahmaza M, Talbi M. [Variation of cognitive functions and glycemia during physical exercise in Ramadan fasting]. Rev Neurol (Paris) 2010;166:721–726.French.

25. Blatter K, Cajochen C. Circadian rhythms in cognitive performance: methodological constraints, protocols, theoretical underpinnings. Physiol Behav 2007;90:196–208.


26. Horne JA, Östberg O. A self-assessment questionnaire to determine morningness-eveningness in human circadian rhythms. Int J Chronobiol 1976;4:97–110.

27. Smith CS, Reilly C, Midkiff K. Evaluation of three circadian rhythm questionnaires with suggestions for an improved measure of morningness. J Appl Psychol 1989;74:728–738.


28. Adan A, Almirall H. Horne & Östberg morningness-eveningness questionnaire: a reduced scale. Pers Individ Differ 1991;12:241–253.

29. Wechsler D. Wechsler Adult Intelligence Scale: technical and interpretive manual. (4th ed). San Antonio, TX: Pearson Assessment, 2008.
30. Egeland J. Measuring working memory with digit span and the letter-number sequencing subtests from the WAIS-IV: too low manipulation load and risk for underestimating modality effects. Appl Neuropsychol Adult 2015;22:445–451.


31. Gignac GE, Reynolds MR, Kovacs K. Digit span subscale scores may be insufficiently reliable for clinical interpretation: distinguishing between stratified coefficient alpha and omega hierarchical. Assessment 2019;26:1554–1563.



32. Crowe SF. Does the letter number sequencing task measure anything more than digit span? Assessment 2000;7:113–117.



33. Cox CS, Chee E, Chase GA, Baumgardner TL, Schuerholz LJ, Reader MJ, et al. Reading proficiency affects the construct validity of the stroop test interference score. Clin Neuropsychol 1997;11:105–110.

34. Golden CJ. Stroop Color and Word Test: a manual for clinical and experimental uses. Wood Dale, IL: Stoelting Co, 1978.
35. Pezzuti L, Rossetti S. Letter-number sequencing, figure weights, and cancellation subtests of WAIS-IV administered to elders. Pers Individ Differ 2017;104:352–356.

37. Jensen AR. Chronometric terminology and paradigms. Jensen ARClocking the mind: mental chronometry and individual differences (1st ed). Amsterdam, Elsevier, 2006, p. 11–41.
39. Hindmarch I. Critical flicker fusion frequency (CFF): the effects of psychotropic compounds. Pharmacopsychiatry 1982;15:44–48.

40. Lotfi S, Madani M, Abassi A, Tazi A, Boumahmaza M, Talbi M. CNS activation, reaction time, blood pressure and heart rate variation during ramadan intermittent fasting and exercise. World J Sports Sci 2010;3:37–43.
41. Lotfi S, Zerdani I, Elouafi L. Learning from a cognitive neuroscience perspective. In: New science of learning. Leiden: Brill, 2023, p. 101–126.
42. Hartley LR, Shirley E. Color-name interference at different times of day. J Appl Psychol 1976;61:119–122.


43. Manly T, Lewis GH, Robertson IH, Watson PC, Datta A. Coffee in the cornflakes: time-of-day as a modulator of executive response control. Neuropsychologia 2002;40:1–6.


44. Jacobson J, Matthaeus L. Athletics and executive functioning: how athletic participation and sport type correlate with cognitive performance. Psychol Sport Exerc 2014;15:521–527.

45. Evansová K, Červená K, Novák O, Dudysová D, Nekovářová T, Fárková E, et al. The effect of chronotype and time of assessment on cognitive performance. Biol Rhythm Res 2022;53:608–627.

47. Johnson MP, Duffy JF, Dijk DJ, Ronda JM, Dyal CM, Czeisler CA. Shortterm memory, alertness and performance: a reappraisal of their relationship to body temperature. J Sleep Res 1992;1:24–29.


48. Monk TH, Carrier J. Speed of mental processing in the middle of the night. Sleep 1997;20:399–401.


49. Monk TH, Buysse DJ, Reynolds CF 3rd, Berga SL, Jarrett DB, Begley AE, et al. Circadian rhythms in human performance and mood under constant conditions. J Sleep Res 1997;6:9–18.



-
METRICS

-
- 0 Crossref
- 1 Scopus
- 1,520 View
- 38 Download
- Related articles in Chronobiol Med