Sleep Quality of Mothers of Premature Infants after Neonatal Intensive Care Unit Discharge
Article information
Abstract
Objectives
Advanced clinical management has increased the survival rate of premature infants. Mothers of premature infants have more concerns about their infant’s health, which may impair their sleep quality. This study aimed to investigate the sleep quality of Korean mothers of premature infants after neonatal intensive care unit (NICU) discharge.
Methods
Face-to-face interviews were conducted for 90 mothers of infants corrected aged 0–12 months who were born at <37 weeks of gestation and had been discharged from the NICU. The questionnaire consisted of the general background of mother including family, employment status and medical history of infants; Pittsburgh Sleep Quality Index (PSQI) to assess the mother’s sleep status; the Brief Infant Sleep Questionnaire to assess the infant’s sleep status.
Results
The mean global PSQI score was 8.0±4.0. In total, 70/90 mothers were identified as poor sleepers (PSQI scale >5). There was significant difference in premature infants’ age, corrected age, weight, duration since NICU discharge, and frequency and duration of night waking. However, it did not show significance in maternal factor, familial background, medical condition of infant.
Conclusion
Most Korean mothers of premature infants experienced poor sleep quality even after their infants were discharged. We found that mothers with good sleep quality had premature infant of older age and more body weight and longer duration since NICU discharge. These findings indicate that at least in the early stages of discharge of premature infant, sleep education for the mother of the premature infant is necessary.
INTRODUCTION
In recent decades, the rates of preterm birth (born before 37 weeks of gestation), the most frequent cause of infant mortality, have increased worldwide, including in Korea [1,2]. Although the survival of premature infants is improving because of advances in clinical management, premature infants are physiologically and developmentally immature and are at a higher risk of morbidity and mortality than full-term infants [3,4].
Mothers of premature infants have more concerns about their infant’s health and feel that their caregiving burden is greater after infants go home, compared to mothers of full-term infants. The majority of mothers described premature birth as a traumatic stressor, which may lead to various emotional responses such as depression and insomnia, regardless of significant complications [5]. Swedish parents have reported negative effects on their sleep during their infants’ hospitalization in the neonatal intensive care unit (NICU) because of anxiety-induced stress [6]. These stress and depression may negatively influence sleep. Importantly, mothers of premature infants may not achieve the sleep quality necessary for adequate daytime functioning.
Among the subjective methods of sleep quality, the Pittsburgh Sleep Quality Index (PSQI), a self-reported questionnaire used worldwide, measures sleep quality during the previous month. The PSQI has been translated and validated in many languages, including Korean, and is used in many clinical sleep studies [7,8]. Sleep quality have been influenced by the sociocultural factor as well as individual factors such as infant’ condition or familial background. However, limited studies have assessed maternal sleep quality after NICU discharge in Korea.
The aim of this study is to investigate the sleep quality of Korean mothers of premature infants after NICU discharge.
METHOD
Face-to-face interviews were conducted for 90 mothers of premature infants corrected age 0–12 months old who were born before 37 weeks of gestation and had been discharged from NICU.
Study subjects
This study was conducted at three tertiary hospitals from August to November 2019.
The questionnaire consisted of three part: 1) the PSQI to assess the mother’s sleep status; 2) the Brief Infant Sleep Questionnaire (BISQ) to assess the infant’s sleep status; and 3) questionnaires to assess the infant’s current age, gestational age, corrected age, cohabitating family members, birth order, age at NICU discharge. Serious medical conditions during hospitalization and complications after NICU discharge were obtained from medical record. Serious medical conditions included sepsis, need for cardiopulmonary resuscitation, grade 3–4 intraventricular hemorrhage (IVH), hydrocephalus surgery, severe retinopathy of prematurity (ROP), necrotizing enterocolitis surgery, inguinal hernia surgery, patent ductus arteriosus surgery, severe infection, bronchial pulmonary dysplasia (BPD), hypoxic ischemic encephalopathy (HIE). Sequelae of prematurity included growth retardation, insufficient oral intake, BPD, severe IVH, HIE, ROP, symptoms of genetic disorders, osteomalacia, and rickets. For twin mother, one questionnaire was asked.
This study was approved by the ethics committee of Korea University Ansan Hospital (No. 2019AS0178). Written informed consent was obtained from all participants.
Assessment of infant sleep using the BISQ
The BISQ includes 11 subjective and objective infant sleep questions about nocturnal sleep duration, daytime sleep duration, number of night awakenings, duration of nocturnal wakefulness, nocturnal sleep onset time, settling time, method of falling asleep, sleep location, and preferred body position [9]. The mothers were instructed to refer to the infant’s sleep patterns during the previous week. In the case of twin, sleep questionnaire for smaller infants was asked.
Assessment of maternal sleep using the PSQI
The PSQI is a widely used and well-validated self-reporting tool that assesses sleep disturbances in adults. The 19-item PSQI measures seven components: subjective sleep quality, sleep latency, sleep duration, habitual sleep efficiency, sleep disturbances, sleeping medication use, and daytime functioning. It is designed to use the total score added by converting the score for each area. The total score ranges from 0 to 21 points, and the higher the score, the lower the quality of sleep. According to the criteria of Buysse et al. [10], this study used the established cutoff a score of >5 to depict poor sleep quality. In this study, Korean version was used [7].
Statistical analysis
Statistical analysis was performed using SPSS for Windows (version 25.0; IBM Corp., Armonk, NY, USA). To compare two group means, the independent two-sample t-test or Mann-Whitney test was used with Bonferroni correction. Further, analysis of variance or the Kruskal-Wallis test was used to analyze the equality of the means for more three groups. The chi-square test was used to analyze the association between good sleep (PSQI score ≤5) and bad sleep (PSQI score >5).
Multiple logistic regression analysis was used to estimate effect of various factors on the mother’s sleep quality. Results are presented as odds ratios (OR) and 95% confidence intervals (95% CI). p-values <0.05 were considered statistically significant.
RESULTS
Demographic characteristics
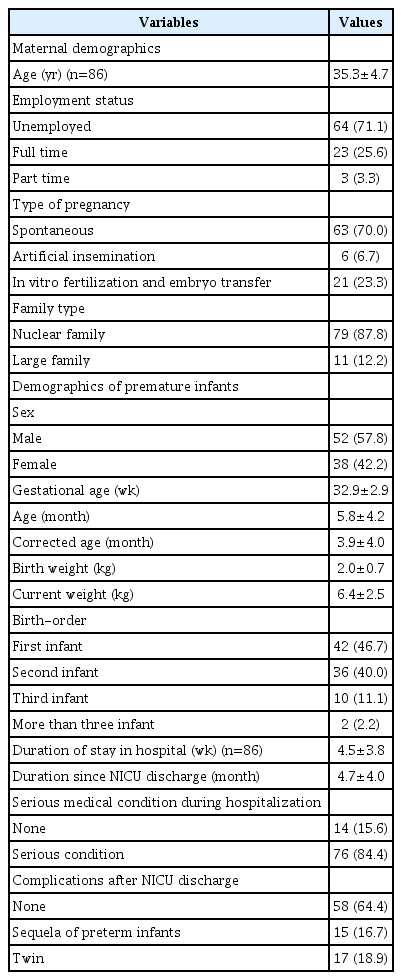
The study included 90 mothers with a mean age of 35.3±4.7 years (range, 22–50 years).
The 90 premature infants had a mean gestational age of 32.9 ± 2.9 weeks (range, 25.14–36.86 weeks); 52 (57.8%) were boys. Their mean corrected age at interview was 3.9±4.0 months (range, -1.43–12.09 months) and the mean duration since discharge was 4.7±4.0 months (range, 0.1–12.8 months). The demographic characteristics of the mothers and the clinical information about their premature infant are presented in Table 1.
Maternal sleep assessment
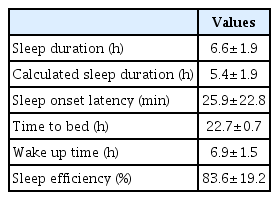
The mean sleep duration was 6.6±1.9 hours, the mean sleep latency was 25.9±22.8 minutes, and the mean sleep efficacy was 83.6±19.2%. The sleep duration reported by mother was different from the sleep duration calculated by went to bed time, waking time, and sleep latency (6.6±1.9 vs. 5.4±1.9 hours). Maternal sleep characteristics are present in Table 2.
Maternal sleep quality in PSQI scores
The mean global PSQI score was 8.0±4.0 (range, 1–17). Each component of PSQI are presented in Table 3.
In total, 70 of 90 (77.8%) mothers were poor sleepers according to the PSQI (score >5). There were significant differences between mothers with good sleep quality and poor sleep quality in infant’s age, corrected age, current weight, duration since discharge, and frequency and duration of night waking (Table 4). However, there were no significant differences in gestational age, familial background including family type, number of children, maternal employment status. In addition, there were no differences in infant sleep status, serious medical condition during hospitalization, and sequelae of prematurity.
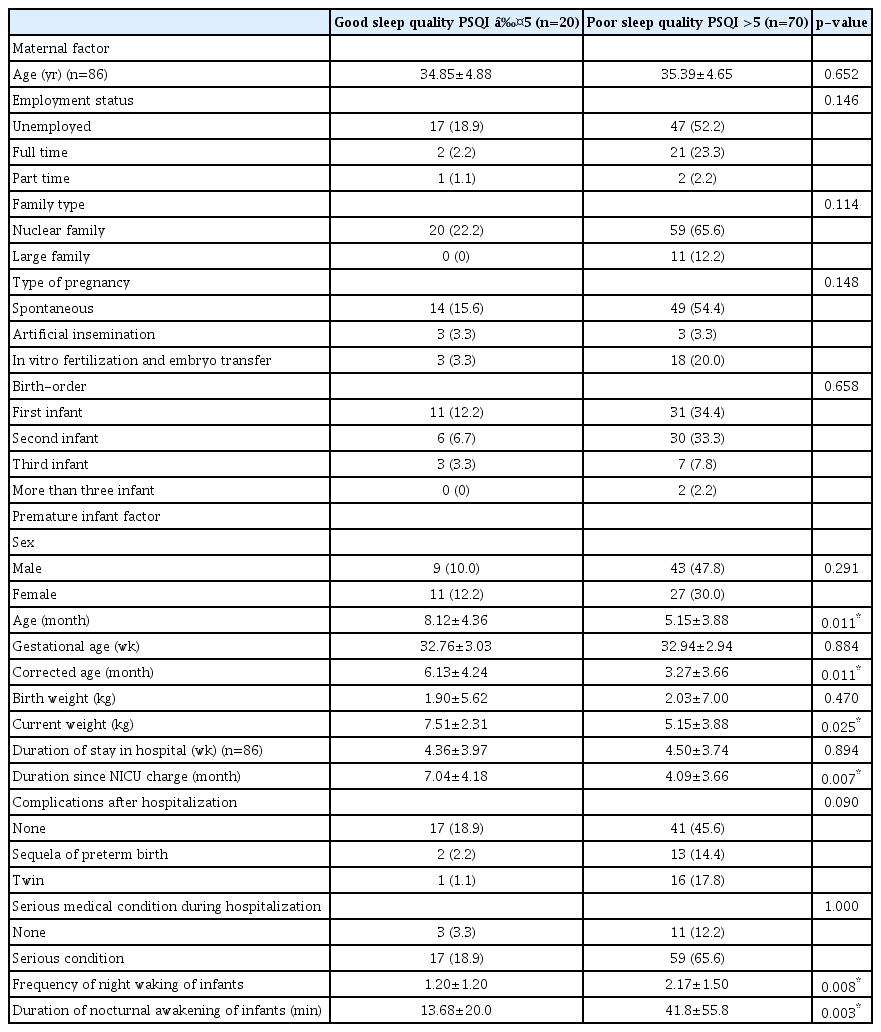
Comparisons of characteristics between good sleep quality and poor sleep quality of the mothers of premature infant according to the PSQI score (n=90)
In multiple logistic regression analysis, maternal sleep quality was related to corrected age of premature infant (p=0.034, OR 0.85, 95 CI 0.74–0.99).
DISCUSSION
In this study, 77.8% of mothers of premature infants had poor sleep quality according to the PSQI. Mothers of premature infants with younger current and corrected ages, lower current weight, shorter duration since discharge, and more night waking during sleep had worse maternal sleep quality (p<0.05). There were no significant differences in gestational age, family type, number of children, serious medical conditions during hospitalization, or sequelae of prematurity. In this study, we found that maternal sleep quality was more closely related to premature infant age and weight than the familial background or complications related to prematurity. To our knowledge, this is the first study to investigate the sleep quality in Korean mothers of premature infants after NICU discharge.
In this study, the mean sleep duration by reported mothers was 6.6±1.9 hours a day and the mean sleep latency was 25.9±22.8 minutes. And calculated sleep duration by went to bed time, waking time and sleep latency was 5.4±1.9 hours. This result is less than the mean sleep duration of 7.7 hours for Korean mothers of 0–6 aged children reported in other study [11]. Even in healthy adults, partial sleep deprivation of 5–6 hours of sleep duration has been shown to result in daytime sleepiness and a negative mood [12]. When it continues for 1–2 weeks, it can lead to cognitive impairment [13]. These mental effects of poor sleep quality negatively affect the mothers’ well-being, which negatively affects their infant [14]. Therefore, sufficient sleep is important for ensuring quality care for premature infants. On regarding the difference between reported sleep duration and the calculated sleep duration, mothers of premature infant may not realize that their actual sleep duration is shorter. This indicated that mothers may not aware of their sleep status which may worsens the sleep quality. An Australian study have reported that educational intervention is effective in preventing postpartum depression (PPD) and sleep disturbance [15]. Therefore, it is helpful to educate mothers of premature infant about adequate sleep duration and a healthy sleeping environment.
Maternal subjective sleep quality is closely related to maternal depressive symptoms and infant caretaking at night [16]. PPD is common and has serious implications for a mother and her newborn. A possible link between placental corticotrophin-releasing hormone and PPD has been hypothesized, but empirical evidence is lacking [17]. Moreover, when premature infants remain hospitalized for several weeks or months, the potential cumulative effect of poor parent sleep on their ability to retain complex health information and participate in decision making is of particular concern [18]. Studies have shown that parents of premature infants had heightened levels of depression, anxiety, and stress compared to parents of full-term infants [19,20]. Carter et al. [21] have reported a dose-response relationship between the degree of prematurity and depressive symptoms, with higher depression scores in mothers of infants born at <33 weeks of gestation than in mothers of infants born at 33–35 weeks of gestation and mothers of full-term infants.
Stress has been known one of the most important risk factors of impaired sleep. Stress experienced by mothers of an infant in the NICU is concomitantly associated with sleep disturbance, fatigue, depression, and poor quality of life [22,23]. Lee et al. [24] has proposed the following overall stress-sleep-health path; 1) stress would directly contribute to sleep disturbance; 2) disturbed sleep would mediate the effect of stress on fatigue and poor quality of life; and 3) fatigue would mediate the correlation between stress, depression, and sleep disturbance in addition to the direct contribution to adverse quality of life.
Maternal separation anxiety is defined as an unpleasant emotional state that involves feelings of guilt, worry, and sadness that accompany short-term separation from the child [25]. Korean mothers’ feelings about premature infants have reported that emotional distress and concern are large, which can be attributed to cultural background [26]. Higher levels of separation anxiety were associated with oversensitivity to physical proximity at bedtime and more involvement during the course of the night [27,28]. Separation anxiety of mother may be caused by abnormal control of breathing during sleep and sleep apnea of prematurity. In particular, premature infants are at a risk of sleep-related life-threatening events such as sudden infant death syndrome. These psychological causes of mothers would have further poor sleep quality through bidirectional paths.
Infants’ sleep patterns are problematic and include night waking with crying, a common concern for parents of infants. In previous studies, the mean frequency of nocturnal waking of full-term infants was 0.8/night (range, 0–2.9/night); just over half (55%) of the participating mothers reported that their premature infants woke 2.1 times per night for a mean 45.7 minutes per night, which is similar to our findings [29,30]. We found that the frequency and duration of night wakings of premature infants affected the quality of the maternal sleep. Frequent nocturnal waking negatively impacts infant and family functioning [31,32]. Also, maternal sleep quality may cause problems with parenting quality. King et al. [33] have reported that mothers with poorer sleep continuity have decreased sensitivity toward their infants.
The findings of abovementioned studies support those of our study—77.8% of mothers of premature infants experienced poor sleep quality. However, it is not yet known how long the premature infant’s mother would have poor sleep quality. We found significant differences in duration since NICU discharge (7.04±4.18 vs. 4.09±3.66 months, p<0.05). This finding is consistent with those of a study that reported a lower rate of night waking at 5 months of age among infants after NICU discharge compared with a term controls; however, by 25–45 months of age, there were no differences [34]. O’Brien et al. [35] have reported that there is no statistically significant decrease in depression score after discharge. However, one study has reported that mothers of premature infants had higher parental stress scores than mothers of term infants at 1–2 years of age and that mothers of very low birth weight premature infants had behavioral problems at 3 years of age [36]. These results including our finding suggest that better sleep quality can be expected as premature infant grows.
Assessment methods of sleep in infants and adults can be subjective or objective. Subjective methods include sleep diaries, self-reported questionnaires, and structured interviews. There are many questionnaires for child and adult, some of which are validated such as the BISQ and PSQI, that assess sleep duration, onset time, and sleep latency in the sleeping environment. The PSQI, one of the most widely used measurement tools for assessing sleep status in adults, is recognized for its reliability and validity in several languages, including Korean [7]. The PSQI has the advantage of calculating total and seven area-specific scores, and cut-off points of global PSQI scores are reported as 5 points; hence, PSQI scores in sleep studies can be cross-culturally compared. In addition, sleep state and quality can be evaluated in more detail and have explanatory power through analysis of the sleep measurement dimension for each of the seven areas [37]. However, the method proposed is complicated; hence, it is difficult and time-consuming to obtain a sleep score.
This study has some limitations. We used self-reported questionnaires of infant and maternal sleep and not an objective tool. A previous study has suggested that infant-induced maternal sleep disturbances resulting from frequent infant nocturnal waking might be more relevant to the mother’s perceived sleep quality than the total amount of time she spent awake at night [16]. Therefore, regarding sleep quality related to subjective feelings such as stress, studies using subjective methods such as self-reported questionnaires may be better reflected than studies using objective methods. Further studies should include objective and subjective methods for assessing sleep quality. Another, sleep quality may be influenced by many other factors such as mental problem and socioeconomic status that were not identified or controlled for in this study. Especially, depression and anxiety are well-known factor that may also require future research.
In conclusion, most mothers of premature infants experienced poor sleep quality even after their infants were discharged from the NICU. We found that mothers with good sleep quality had older age and more body weight and longer duration since NICU discharge. These findings indicate that at least in the early stages of discharge of premature infant, sleep education for the mother of the premature infant is necessary.
Acknowledgements
This research was supported by a grant of the Korea Health Technology R&D Project through the Korea Health Industry Development Institute (KHIDI), funded by the Ministry of Health & Welfare, Republic of Korea (Grant number: HI19C0273).
Notes
The authors have no potential conflicts of interest to disclose.
Author Contributions
Conceptualization: Won Hee Seo. Formal analysis: Jaehyung Cha. Investigation: Eui Kyung Choi, Jeong Hee Shin. Methodology: Byung Min Choi, Eui Kyung Choi, Jeong Hee Shin, Kyu Hee Park, Jang Hoon Lee. Supervision: Won Hee Seo. Writing—original draft: Dae Seong Kim. Writing—review & editing: Byung Min Choi.