Brain Activation Changes in Insomnia: A Review of Functional Magnetic Resonance Imaging Studies
Article information
Abstract
Numerous studies have examined factors associated with the occurrence of insomnia. With recent advances in brain imaging studies, neurobiological substrates of insomnia have received attention. In this review, we explore changes in brain activity in insomnia and their clinical and neurobiological interpretation by examining previous functional magnetic resonance imaging (fMRI) studies of insomnia. First, the question of whether insomnia may be related to attentional bias toward sleep-related stimuli has been an issue. Although controversies persist, previous fMRI studies have supported attentional bias to sleep-related stimuli in insomnia patients. Second, many studies have investigated which brain regions are involved in impaired cognitive performance of insomnia using fMRI. The prefrontal cortex or caudate nucleus has been implicated in cognitive impairment with insomnia. Third, we focused on the question of which brain regions are involved in emotional dysregulation in insomnia and its origins. Previous studies using fMRI suggested that sleep disturbance may occur due to abnormal brain function, such as reduced functional connectivity between the amygdala and other areas. Besides these factors, a variety of interconnected factors may have an impact on sleep disturbances, especially the interplay of stress and insomnia; this requires further research. Future fMRI studies of insomnia based on various theoretical models or novel imaging technologies might reveal the neurobiological pathways underpinning the development and maintenance of insomnia.
INTRODUCTION
Sleep disorders are becoming pervasive in modern society. According to a survey conducted by the National Institutes of Health, about 50–70 million Americans suffer from sleep disorders, reporting sleep-related symptoms that could degrade the quality of their lives. Among various sleep disorders, insomnia is regarded as the most common.
Patients suffering from insomnia have been suggested to have impaired neurobiological function compared with those without insomnia. In particular, neurobiological dysfunction in cognitive and emotional processes have been suggested as principal characteristics of insomnia [1-5]. The relationship between insomnia and neurobiological dysfunction might be explained by specific neuroscientific theories. According to the hyperarousal theory, patients with insomnia become obsessed with sleep-related thoughts that flash through their minds, and they may fail to control excessive worry and rumination regarding poor sleep and its consequences [6-9]. These cognitive processes could result in physiological hyperarousal [9,10]. Subsequent cortical hyperactivity may then interfere with sleep. Thus, patients are trapped in a vicious cycle of torment in which psychological and biological hyperarousal continues to aggravate their sleep disturbance and vice versa. Many studies based on a range of theories explaining insomnia have reported that insomnia symptoms are correlated with specific patterns of brain activity.
In the current review, we investigated differences in brain activity patterns between patients with insomnia and good sleepers without significant insomnia. Here, we discuss the relationships between insomnia-related brain areas and the various clinical characteristics and etiology of insomnia. For this purpose, we reviewed previous functional magnetic resonance imaging (fMRI) studies that examined specific neural functions and patterns in insomnia. Previous fMRI studies of insomnia have reported three main findings: 1) attentional bias to sleep-related stimuli; 2) deficits in cognitive functioning; and 3) impairment in emotional processing. Although many methodological approaches have been employed to reveal the neurobiology of insomnia, we decided to focus on fMRI studies, which can be conducted during various kinds of tasks and can demonstrate functional changes in specific neurobiological processing in insomnia.
ATTENTIONAL BIAS TO SLEEP-RELATED STIMULI
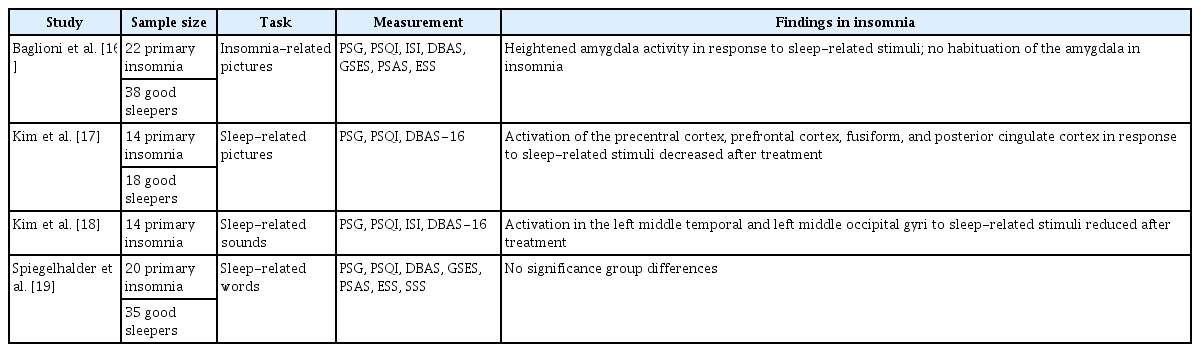
Whether attentional processing in insomnia differs when people are exposed to sleep-related stimuli remains controversial [11]. Attentional bias in insomnia refers to the observation that attention during insomnia tends to be selectively focused on sleep-related content. According to the attention–intention–effort model, attention to sleep-related stimuli is one indicator of the development of chronic insomnia from acute or transitional insomnia [12]. This attentional focus in insomnia reveals patients’ preoccupation with good sleep and their anxiety regarding whether another night of bad sleep lies ahead. To identify the neurobiological process underlying attentional bias to sleep-related stimuli, experiments have presented sleep-related words such as fatigue, sleep-related images such as a picture of a bedroom, or sleep-related sounds such as an alarm clock [13-15]. Attentional bias was measured by assessing reaction times or tracking eye movements.
A few neuroimaging studies tried to investigate neural basis of insomnia with tasks involving sleep-related stimuli (Table 1). The study has revealed hyperactivity in the amygdala when patients with insomnia were exposed to insomnia-related stimuli [16]. Hyperactivity in the amygdala might indicate that the individual recognizes sleep-related stimuli as negative and threatening. The authors also suggested that altered emotional processes in insomnia may be due to a failure to regulate fearful emotions. In addition, hyperactive brain patterns in the precentral cortex, prefrontal cortex, and default mode network in response to sleeprelated stimuli before cognitive behavioral therapy (CBT) for insomnia (CBT-I) were reversed after CBT-I [17]. Furthermore, when exposed to sleep-related sounds, patients with insomnia showed reduced activity in the left middle temporal and left middle occipital gyri after CBT [18]. However, there was no change in brain activation in response to sleep-related words in insomnia patients [19]. Although many studies have reported that subjects had difficulty maintaining attention to a task after sleep deprivation was manipulated [20-22], relatively few have reliably demonstrated attentional bias in insomnia [23]. Thus, whether attentional bias generally occurs in insomnia remains controversial. Moreover, how attentional bias occurs and develops in insomnia remains unclear. Future studies should examine the role of attentional bias in insomnia and determine which neural regions are involved in sleep-related attentional bias.
DEFICITS IN COGNITIVE FUNCTIONING
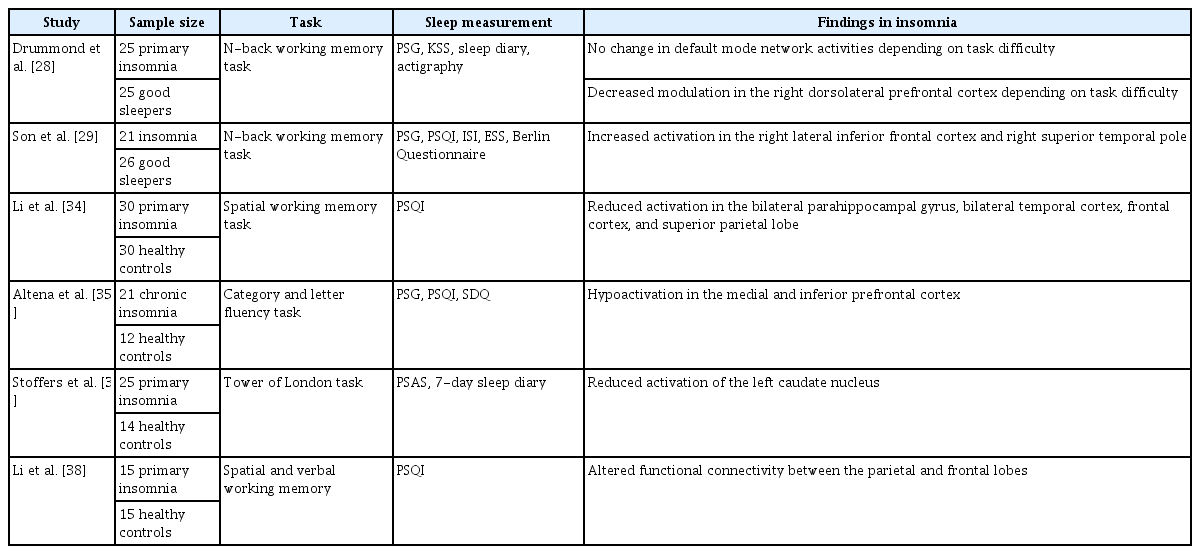
Sleep disturbances can also influence cognitive functioning in patients with insomnia. Patients suffering from insomnia have been reported to exhibit poor performance on cognitive tasks and abnormal activity patterns in the prefrontal cortex and sub-regions known to be involved in higher cognitive functions, such as goaldirected behavior, self-regulation, and working memory [24-27]. A number of studies have explored the brain activity patterns of patients with insomnia while they performed cognitive tasks in a magnetic resonance (MR) scanner (Table 2).
The N-back task, which requires that subjects recall a stimulus presented “n” items earlier, has been used in some fMRI studies to measure working memory and related brain activity patterns in patients with primary insomnia [28,29]. The difficulty of the Nback task depends on the number of items that must be remembered in order. Previous fMRI studies have reported that the more difficult a cognitive task is, the less active the default mode network becomes [30-32]. However, in subjects with primary insomnia, there were no change in default mode network activity regardless of task difficulty [28]. This suggests that those with primary insomnia may fail to inhibit task-irrelevant functioning during goaldirected performance. This finding might be related to the sleep loss of insomnia patients, as people with sleep deprivation were reported to be vulnerable to modulation of the default mode network [33].
Furthermore, decreased activation among insomnia patients has been reported in brain regions that are normally active during cognitive processing. For example, decreased activation in insomnia patients has been reported in the bilateral frontal cortex, bilateral temporal cortex, and bilateral para-hippocampal gyrus during performance of a spatial working memory task [34]. Decreased prefrontal activation during a verbal fluency task has also been reported in insomnia, although there was no difference in behavioral performance between those with chronic insomnia and the control group [35].
In addition, similar to patients with damage to the ventral medial prefrontal cortex, patients with primary insomnia showed poorer performance on the Iowa gambling task assessing decision-making ability [36]. The above findings suggest that patients with insomnia could be vulnerable to making irrational decisions under uncertainty, and their decision-making difficulty may be related to prefrontal dysfunction.
Insomnia patients also showed reduced activation in the left caudate nucleus during cognitive tasks [37]. High scores on the hyperarousal scale predicted reduced activation in the caudate region. These findings may suggest abnormalities in arousal regulation and executive function.
Altered frontal–parietal network connectivity during spatial and verbal working memory tasks was also reported in insomnia patients [38]. This finding was interpreted as indicating that disruption of the frontal–parietal network may lead to difficulty in memory retrieval in insomnia patients.
IMPAIRMENT IN EMOTIONAL REGULATION
Sleep plays an important role in regulating emotion. Sleep disturbances such as insomnia disrupt appropriate control over emotions. People with cumulative sleep loss are likely to experience negative experiences with greater intensity [39,40]. One study found that the dorsal anterior cingulate cortex (a part of the limbic system) was activated in response to both past and novel emotional distress in patients with insomnia [41]. This finding indicates that insomnia or related sleep disturbances may interfere with the process of resolving past stressful or painful memories. Another finding also suggests that patients suffering from insomnia have altered emotional perception [4]. Moreover, a few neuroimaging studies investigated which brains are involved in emotional dysregulation in insomnia (Table 3).
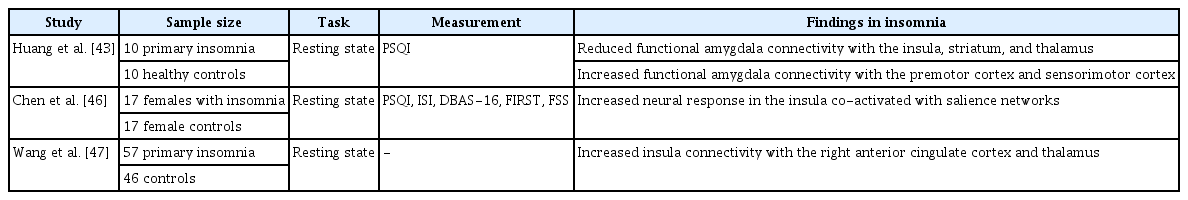
Findings on the connectivity of brain circuits that are deeply involved in emotional regulation also suggest emotional circuit dysfunction in insomnia patients. The amygdala has been regarded as a core regulator of emotional circuits [42]. One study investigated how the functional connectivity of the amygdala works in primary insomnia by analyzing resting-state connectivity [43]. This study reported that patients with insomnia showed reduced functional connectivity of the amygdala with the insula, striatum, and thalamus. In contrast, insomnia was associated with increased functional connectivity of the amygdala with the pre-motor cortex and sensorimotor cortex. Moreover, this connectivity was positively correlated with sleep disturbances as assessed by the Pittsburgh Sleep Quality Index. The amygdala is known to prepare the body for confronting or avoiding dangerous situations (fight or flight) [44]. Therefore, these results could be interpreted as showing that insomnia may induce a state of internal threat even during rest. This abnormally increased sensation of threat could contribute to the daytime fatigue that characterizes insomnia. However, another study showed that self-reported sleep loss was negatively correlated with resting-state functional connectivity of the left amygdala with the frontal cortex [45]. These results demonstrate that under poor sleep conditions, individuals lose the ability to process and regulate their emotions due to reduced connectivity between the amygdala and frontal cortex. The role of connectivity between the amygdala and frontal cortex in insomnia patients remains controversial and requires additional well-controlled studies with a more clearly defined insomnia sample.
Other studies have examined the role of the insula in insomnia [46,47]. fMRI and electroencephalogram results revealed an increased neural response in the insula that was co-activated with the salience network in insomnia [46]. Increased connectivity in insomnia patients was also reported between the insula and emotional circuits, including the right anterior cingulate cortex and thalamus [47]. These finding raise the possibility that this insula–salience network co-activation may reflect psychological distress, as seen in the worry and obsessive rumination commonly found in chronic insomnia patients.
STRESS-INDUCED INSOMNIA
Stress and sleep are associated in a complicated way. People might experience sleep problems, especially insomnia, when they confront socially, psychologically, or physically stressful situations. Excessive stress has been suggested to trigger sleep disturbances such as insomnia [48]. Stress can also play a role in the transition from transient insomnia to chronic insomnia. In keeping with theories about the relationship between stress and sleep, previous studies have found that the response of rats to stress is similar to that in stress-induced insomnia [49]. Moreover, neural activity in the amygdala during exposure to negative stimuli predicted perceived stress in patients with insomnia [50]. One study reported a range of factors moderating and mediating the relationship between stress and insomnia, including cognitive intrusion, self-distraction, behavioral disengagement, and substance use [51]. Numerous studies have examined the psychological relationship between stress and insomnia. However, despite the important relationship between stress and insomnia, to the best of our knowledge, no fMRI study has explored the interaction among stress, insomnia, and its neurobiological substrates. Further study is needed to investigate the involvement of neural circuits in the interplay between stress and insomnia.
CONCLUSION
Previous fMRI studies have demonstrated altered brain activities in patients with insomnia compared with healthy good sleepers. Altered brain activities among insomnia patients have been explained in various ways, including attentional bias to sleep-related stimuli, deficits in cognitive functioning, and impairment in emotional processing.
As mentioned earlier, future brain imaging studies of insomnia may examine the brain activity underlying stress-induced or stressrelated insomnia. Several other issues would also be good candidates for future studies. Investigations of changes in the sleep–wake cycle or circadian rhythm of insomnia patients may be needed. One study reported that abnormal neurobiological circuits associated with sleep–wake regulation can induce transient awakening, one of the main symptoms of insomnia [52]. Sleep-state misperception, which is commonly found in insomnia patients, would also be a good target for brain imaging studies. Importantly, future neuroimaging studies could reveal the neurobiological substrates of each of the factors contributing to sleep-state misperception in insomnia [53]. Future studies could also help to conceptualize the roles of bottom-up and top-down processes in primary insomnia [54].
Additional studies are also needed to synthesize various theoretical models and reveal the complex mechanisms underlying insomnia in a way that considers the interactions between psychological and biological factors. Furthermore, to relieve the dysfunctional symptoms resulting from insomnia, research needs to explore the fundamental neural circuits involved in the development and maintenance of insomnia. Investigations with sophisticated experimental designs and highly advanced imaging techniques may also be necessary.
Acknowledgements
This study was supported by the Brain Research Program through the National Research Foundation of Korea, funded by the Ministry of Science, ICT & Future Planning (No. 2016M3C7A1904338).
Notes
The authors have no potential conflicts of interest to disclose.
Author Contributions
Conceptualization: Yunjee Hwang, Seog Ju Kim. Data curation: Yunjee Hwang, Seog Ju Kim. Formal analysis: Yunjee Hwang. Funding acquisition: Seog Ju Kim. Investigation: Yunjee Hwang. Methodology: Yunjee Hwang. Project administration: Seog Ju Kim. Resources: Seog Ju Kim. Software: Seog Ju Kim. Supervision: Seog Ju Kim. Validation: Seog Ju Kim. Visualization: Yunjee Hwang. Writing—original draft: Yunjee Hwang. Writing—review & editing: Seog Ju Kim.